The status quo and advances in categorization of congenital cataract
关键词
摘要
Congenital cataract (CC) is one of the most common causes of pediatric visual impairment. As our understanding of CC's etiology, clinical manifestations, and pathogenic genes deepens,various CC categorization systems based on diferent classifcation criteria have been proposed. Regrettably, the application of the CC category in clinical practice and scientifc research is limited. It is challenging to obtain preciseinformation that could guide the timely treatment decision-making for pediatric cataract patients or predict their prognosis from a specific CC classification. This review aims to discuss the statusquo of CC categorization systems and the potential directions for future research in this field, focusingon categorization principles and scientific application in clinical practice. Additionally, it aims to propose the potential directions for future research in this domain.
全文
INTRODUCTION
THE PRINCIPLE OF CATEGORIZATION AND ITS DEVELOPMENT PROGRESS
By far, there has been no unified standard for the categorization of CC. In the early stage, specifc cataracts were described based upon the name of investigating doctor or the patients’pedigree, such as Marner cataract[7]and Coppock cataract[8]. In 1942, Adamas frst described Coppock cataract as a circular spotted disc in the center of the lens under slit-lamp examination.[9] Subsequently, scholars began to classify CC based on the morphology andanatomical locations of lens opacities.[10] Meanwhile, in pursuit of understanding the pathogenesis of cataracts and pursuing etiological treatment, the etiology of CC was also used as a classification criterion. With the advancement of medical technology, innovative approaches such as genome-wide arrays, whole genome sequencing technologies, and advanced ocular examinations had been employed to classify CC. Gene sequencing technology facilitated the exploration of the relationship between causative genes and cataracts, and scholars triedto classify CC based on the correlation between genotype and phenotype.[11] Recently, advancements in ultrasonic and optical measurement technology have enabled the identification of more detailed characteristics of the anterior segmentby various measuring instruments.[12-14] Classifcation systems for CC based on the outcomes of anterior segment measuring instruments have emergedto investigate the association between CC and other anterior segment abnormalities.Obviously, current categorization systemsfor CC are complexity and diversity, due to diferent principles of classification (Figure 1). From the perspective of ophthalmologists, these sytems possess both strengths and weaknesses and their respective areas of application are diferent.
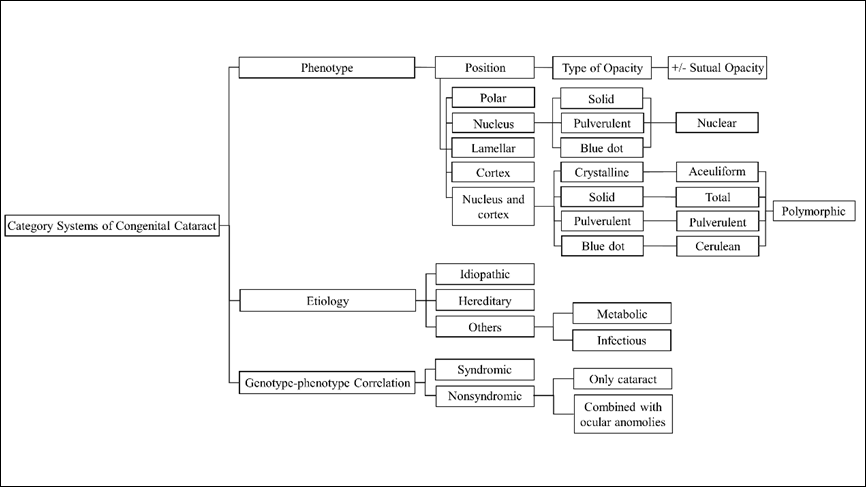
Figure 1 Diffirent categorization systems of congenital cataract
CATEGORIZATION SYSTEM BASED UPON MORPHOLOGY AND LOCATIO
The morphology and locations of CC exhibit considerable variation.[15] In terms of morphology, CC can be classified as solid, pulverulent, or blue dot, among others. With respect toanatomical locations, CC can be categorized as nuclear, cortical and more, as depicted in Figure 2. Multiple classifications have been the morphology or locations of CC.[4,15-19] Reddy et al.[20] presented a relatively comprehensive classifcation method by integrating the morphology and locations of opacities within the lens. According to this classifcation, CC is divided into anterior and posterior polar, nucleus, lamellar, nucleus and cortex, and cortex based on their repective locations. Among these, the nucleus and cortex can be further subcategorized into solid, pulverulent, blue dot and crystalline based on the type of opacity. This classification system is widely employed in clinical practicedue to its convenience in facilitating directdiferential diagnosisby ophthalmologists.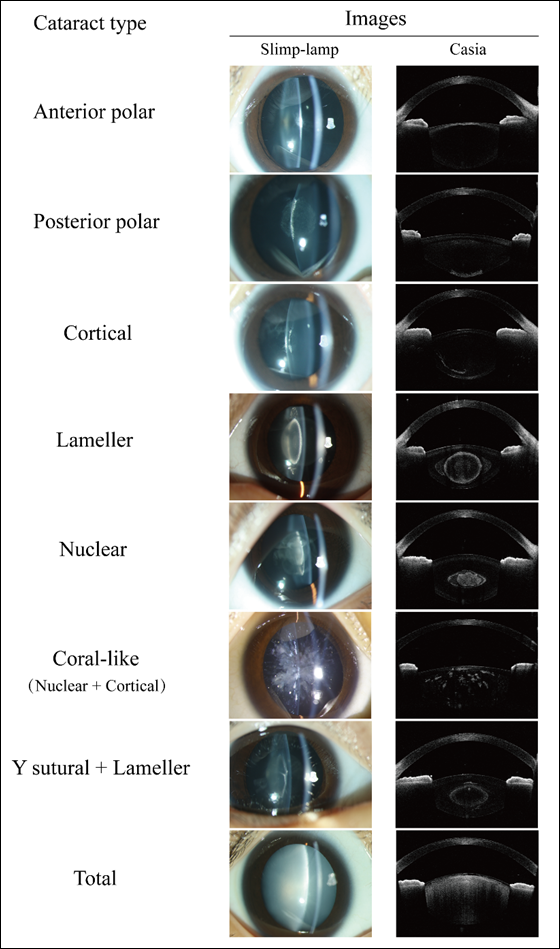
Figure 2 Slit-lamp and Swept-source anterior segment Optical Coherence Tomography (AS-OCT, Casia) images of different congenital cataract morphologies
The morphology of cataracts plays an important role in predicting surgical outcomes. Marshall et al.[21] observed significant variations in that postoperative visual acuity among different cataract types, with lamellar and posterior lenticonus groups exhibiting the most favorable outcomes, while nuclear group demonstrated relatively poorer results. Li et al.[24] documented suboptimal visual outcomesin patients with bilateral total congenital cataracts, with 93/176 (52.8%) eyes achievinga postoperative best-corrected visual acuity (BCVA) of < 20/200. Kim et al.[25] found that postoperative BCVA for posterior polar cataracts (0.34±0.12 logMAR) surpassedthat of nuclear (0.39±0.22 logMAR) and total (0.58±0.42 logMAR) cataracts. Mistr et al.[26] reported surgery outcomes of 30 eyes with posterior polar cataracts and 32 eyes with posterior lenticonus, revealing that 84% and 68% of eyes in the respective groups achieved acuities better than 20/40. Chen et al.[27] reported similar findings, with 21/30 (70%) of eyes diagnosed with posterior polar cataracts or lenticonus achieving a BCVA better than 20/40. Conversely, Kekunnaya et al.[28] found poorer visual outcomes in cases of posterior lenticonus, with 29/59 (49%) of eyes exhibiting a BCVA worse than 20/100 or eccentric fixation 6 months postoperatively. Generally, total and nuclear cataracts are related to unsatisfactory visual prognosis, requiring aggressive surgical intervention as early as possible. The varying prognoses observed in posterior polar cataracts and lenticonus indicate that the influence of other factors such as age at surgery, potential fundus lesions and preoperative visual acuity.[27-28] Treatment strategies should be tailored according to the specifc morphology of cataract.
While this classification is a simple and useful tool in clinical practice, it is not without its drawbacks. Firstly, due to the absence of a detailed classification standard, ophthalmologists may encounter difficulty in precisely discerning the specific locations or morphology of CC sometimes. Furthermore, this classification seems to overlook other coexisting ocular conditions. Common ocular comorbidities in congenital cataracts include aniridia, microphthalmos, persistent fetal vasculature (PFV), nystagmus, and strabismus. Nagamoto et al.[23] found that compared to unilateral cases, bilateral cases exhibited a notablyhigh frequency of nystagmus, while in unilateral cases, strabismus emerged as the most prevalent associated ocular diseases. Additionally, strabismus, nystagmus and microphthalmos were frequently observedin cases of total cataract, while PFV often presenting in eyes afected by posterior subcapsular/polar cataracts. Haargaard et al.[6] highlighted the frequent occurrence of PFV in unilateral cases, while noting that microphthalmos and microcornea were more prevalent in bilateral cases. Ocular comorbidities hold the potential to influence the formulation of treatment strategies and patients’prognosis. For example, in cases of PFV, the fetal vessels may strongly adhere to the posterior capsule, resultingin preoperative or intraoperative posterior capsule rupture[29] and intraoperative hemorrhage[4]. To avoid severe complications, comprehensive preoperative examinations are required, and surgical procedures must be meticulously planned. Futhermore, individuals with CC may also present various multisystem disorders, such as Down’s syndrome[30], Oculo-Facio-Cardio-Dental (OFCD) syndrome[31], Trisomy 21, Lowe syndrome and many others[32], all of which have the potential to signifcantly impact patients’prognosis.
Recently, there have been rapid advancements in anterior segment measurement instruments, such as anterior segment Optical Coherence Tomography or Pentacam connected to a digital Scheimpfug rotational camera. Based on the locations of lens opacities shown in slit-lamp and Pentacam examinations, Liu et al.[33] established a classification for CC into four groups: total, anterior, interior, and posterior cataracts. In this classifcation, total cataract is defned as opacity of entire lens, while interior cataract refers to denotes opacity of the interior lens without involvement of anterior or posterior capsules. Anterior and posterior cataracts indicate lens opacities with involvement of anterior or posterior capsules, respectively. This classification method, being straightforward, is both simple and practical for ophthalmologists to employ in in clinical settings. Using this classification, they explored the relationship between locations of CC and other anterior segmental characteristics. Although signifcant corneal astigmatism (CA) was found in all types of CC, anterior cataract showed the strongest association with CA. As the locations of CC became more posterior, the value of CA decreased. Additionally, anterior chamber depths (ACD) varied across the different types, with interior and posterior cataract had greater ACD, while total and anterior cataracts exhibited shallower ACD. Other studies also focusedon the abnormal anterior segment structure in CC. Han et al.[34] observed the distribution of anterior corneal astigmatism (ACA), posterior corneal astigmatism (PCA), and total corneal astigmatism (TCA) of patients with CC. They also found large CA in patients with CC, with 70.3% of cataractous eyes (378/538) displaying an ACA exceeding1.25 D, and 72.8% of cataractous eyes (392/538) exhibiting a TCA exceeding1.25 D. Among cataractous eyes, 70.6% of cataractous eyes (380/538) had PCA ranging from 0.25 to 0.75 D. In addition to anterior segmental characteristics, Liu and coworkers[35] further into the link between visual function and ocular structure in patients diagnosed with posterior cataract. The discovered that compared with their contralateral healthy eyes, cataractous eyes exhibited a prolonged peak time of P100 of PVEP-60’, along with a diminished amplitude of P100 of PVEP-60’.
In contrast to the traditional 2-dimensional image-based classification, this modified classification system leverages 3-dimensional images for categorization of CC, yielding heightened precision in clinical applications. Furthermore, it facilitates the anticipation of potential anterior segment irregularities based on the locations of lens opacities and ofers more pragmatic guidance in formulating treatment approaches for ophthalmologists. Nonettheless, despite its simplicity and practicality in clinical settings, this method overlooks the varied morphologies and severity gradients of CC, alongside potential ocular or systemic comorbidities. Moreover, in instances where the morphology of CC is intricate, this classifcation method may not be applicable.
CATEGORIZATION SYSTEM BASEDUPON ETIOLOGY
The etiology of cataracts presents a range of causes. Cataracts can be categorized into several types based on their origins, including idiopathic, hereditary, metabolic, infectious cataracts.[36] Reports indicate that idiopathic cataract represent the most prevalent form, accounting for up to 50% of all the cases.[6,37-38] Patients diagnosed as idiopathic CC can have other ocular or systemic disorders, thereby requiring the exclusion of hereditary, infectious and other etiologies. Notably, unilateral cases are more likely to be idiopathic in comparision to bilateral instances. While the risk factors of idiopathic CC remain unconfirmed, earlier reports have suggested a potential correlation with prenatal or perinatal causive factors.[37] Following idiopathic cataracts, hereditary cataract represent the second common type, comprising over 20% of all the cases.[39] Hereditary modes of CC include autosomal dominant, autosomal recessive, and X-linked recessive inheritance, with autosomal dominant inheritance being the most prevalent.[40] Hereditary cataracts demonstrate considerable clinical and genetic heterogeneity.[41-42] Additionally, metabolic disorders such as galactosemia, hypoglycemia, hypocalcemiacan lead to the development of cataracts.Galactosemia arises from a deficiency in any of the three enzymes, including galactokinase, galactose-1-phosphate uridy1 transferase, or uridine diphosphate-galactose-4-epimerase.[43] Galactose is converted into galactitol in the lens, leading to the accumulation of galactitol and subsequent osmotic changes. Initially, the lens exhibitsoil-droplet opacification, which may progress to cortical or nuclear opacification. This process is reversible if galactose is promptly excluded from the diet during the early stages. Additionally, intrauterineinfections, particularly those caused by toxoplasma gondii, rubella virus, syphilis, cytomegalovirus, herpes simplex virus (TORCH), can lead to CC. Among these, rubella virus infection is the most prevalent. Besides cataract, the clinical manifestations of congenital rubella syndrome include glaucoma, pigmentary retinopathy, heart disease, and brain dysfunction. However, the precise pathogenesis of CC resulting fromintrauterine rubella infection remains elusive. Nguyen et al.[44] reported that theviral infection within the ciliary body may underlie the development of cataract.
To a certain extent, classifying cataractsbased on their etiology can serve as a guide for clinicians in determining treatment strategies. Forinstance, patients diagnosed with hereditary CC, may require genetic testing and counseling for their families. Individuals with galactosemia would benefit from a galactose-free diet. In cases where cataracts are caused by rubella virus, clinicians should consider the patients’general health to rule out severe systemic comorbidities. However, achieving the precise etiological diagnosis of CC can be challenging at times. In clinical practice, etiology classification is not used independently but rather combined with other CC classifications to facilitate improved diagnosis and treatment.
CATEGORIZATION SYSTEM BASED UPON GENOTYPE-PHENOTYPE CORRELATION
With the emergence and application of genetic technology, researchers have embarked on the exploration ofpotential genetic abnormalities associated with hereditary cataract. Presently, the literature reports an association of more than 100 genes with both nonsyndromic or syndromic hereditary cataract.[40,45] Hereditary cataracts show significant variation within and between families. It is noteworthy that a single mutation can give rise to diverse froms of cataracts, while identical types of cataracts can result from mutations in diferent genes. To elucidate the the suspected genetic causes based on phenotype, some scholars have categorized the implicated genesinto four groups, guided by the genotype-phenotype correlation: the genes implicated in syndromic cataracts, genes associated specifcally with congenital cataracts within syndromic cases, genes exclusively associated with cataracts, and genes associated with both cataract and eye anomalies.[11]The extent to which currently known genes can account for the occurrence of cataracts has varied across different studies. More than 50 genes have been identified in association with nonsyndromic cataract, including crystalline genes, membrane transport protein genes, developmental regulator and transcription factor genes, cytoskeletal protein genes, and transmembrane protein genes.[46] Crystalline genes are particularly prominent in nonsyndromic hereditary cataracts, demonstratinga range of phenotypicv ariations. Berry et al.[47] reported 10 different crystallin variants in CRYAA, CRYBA1, CRYBB1, CRYGD, and CRYGC, with nuclear or lamellar phenotypes being predominant. A variant in CRYBB2 was found to be associated withnuclear-sutural phenotype.[48] Rogaev et al. indentifed variant in the γ-crystallin gene.[49] that may contribute to non-nuclear phenotypes. Cai et al.[50] discovered avariant in the CRYGD gene associated with coralliform phenotype. Disease-causing variants in CRYAB, CRYBA2, CRYBA3, CRYBA4, and CRYGS have been reported, and these variants exhibit association with various cataract phenotypes.[48,51] Mutations in membrane transport protein genes can result in a diverse array of phenotypes, including total, nuclear, Y-sutural with nuclear, pulverulent, lamellar, lamellar sutural, pearl box, coralliform, punctate, Coppock-like cataracts.[11,52-53] A Variant in LIM2 gene, in particular, has been linked to membranous cataract.[54] Mutation in developmental regulator and transcription factor genes, such as of PAX6 and HSF4, are associated with a wide range of phenotypes, including lamellar, punctate, coralliform, anterior and posterior polar, nuclear cataracts.[52,55] Whereas, variants in PITX3 are associated with narrower range of cataract phenotypes, including posterior polar, total, and nuclear phenotypes.[55] It is worth noting that variants in some of the above-mentioned genes can give rise to other ocular anomalies. For instance, Zin et al.[56] reported that a missense variant in CRYBB3 could lead to both cataract and microphthalmia. Gao et al.[57] found that variant in CRYGD could cause cataract along with nystagmus. When cataracts co-occur with abnormalities in the other organs, it results in syndromic cataracts. More than 200 syndromes have been associated with hereditary cataracts, exhibiting diverse morphologies and genetic causes.[11,46,58]
Undeniably, this classification system has the potential to enhance our understanding of the genotype-phenotype correlation in hereditary cataracts, thereby aiding in the genetic counseling for patients and their families. However, due to its clinical and genetic heterogeneity of the condition, there are challenges in establishing a precise genotype-phenotype correlation of hereditary cataract. Therefore, it is difficult to solely rely on complex phenotypes for gentic classification of hereditary cataracts.
CONCLUSION
There exist multiple categorization systemsfor CC, some of which are utilized in genetic and basic researches. However, the practical application of CC categorization systems in clinical practice is limited, such as appearing as only baseline data in clinical studies or neglecting eye and systemic symptoms other than cataract itself in clinical practice. At present, obtaining precise data on morphology, etiology, and genetics within a specific classification remains challenging, and not all classifcations reliably predict the visual outcomes of patients. Therefore, to comprehensively characterized CC conditions and facilitate in treatment decisions and prognostic assessment, it is important for clinicians to adopt a comprehensive approach by utilizing multiple CC classifications instead of relying solely on a specific one. By incorporating various classifcation systems, clinicians can gain a more complete understanding of the condition, leading to improve management strategis and patient care.
FUTURE RESEARCH
Currently, many CC categorization systems lack the ability to precisely predict the visual prognosis of pediatric cataract patients with varying characteristics. While some studies have reported the postoperative visual acuity based on morphological categorization of different CC types, further research is needed to investigatethe correlation between various CC categorization systems and visual prognosis from multiple clinical perspectives, encompassing factors such as visual acuity, surgical complications and others.In the context of CC classification, slit lamps and anterior segment measurement instruments are commonly used. However, young children often face challenges in cooperating with these examinations. To overcome this issues, the utilization of handheld eye examination instrument during sedation has proven to be a more feasible approach for collecting eye information from young children. The developmetof additional handheld eye examination instruments in the future will further facilitate the classification of CC.
In addition, future research should focus on integrating dimensions for classifying CC. The phenotypic characteristics of CC patients, including ocular biometry of anterior and posterior segments, usually often exhibit variations among individuals. The current CC categorizations ystems primarily rely on single standards, which may not adequately capture the comprehensive ocular characteristics of patients. Therefore, it is important to develop new categorization systems that incorporate multiple dimensions. For example utilizing ocular biometry and considering the conditions of anterior and posterior segments as classification criteria. Furthermore, it is crucial to explore the correlation between this new classifcation system and the visual prognosis of children with CC. By doing so, we can enhance the clinical value and applicability of the classifcation system. The ultimate objective of future research is to improve the clinical and scientific utility of CC categorization systems, leading to more accurate diagnoses, effective treatment approaches, and better outcomes for patients.
Correction notice
NoneAcknowledgement
Our heartfelt appreciation to Ms. Yin Qiuxia for her dedicated eforts in meticulously improving the language and grammar of this article with her precious time.Author Contributions
(I) Conception and design: Zhenzhen Liu(II)Administrative support: Zhenzhen Liu
(III) Provision of study materials or patients: Yingshi Zou and Yunqian Li
(IV) Collection and assembly of data: Yingshi Zou and Yunqian Li
(V) Data analysis and interpretation: Yingshi Zou and Yunqian Li
(VI) Manuscript writing:All authors
(VII) Final approval of manuscript:All authors
Funding
This work was supported by the Joint Funding Project of Municipal Schools (Colleges) of Science and Technology Program of Guangzhou, China (2023A03J0188) and the Guangzhou Municipal Science and Technology Project (202201011815).The funding organizations had no role in the following aspects: design and conduct of the study; the collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and the decision to submit the manuscript for publication.