Prognostic nomogram for patients with primary conjunctival malignant tumors: a study based on SEER data
关键词
摘要
全文
HIGHLIGHTS
INTRODUCTION
Conjunctival tumors are frequently encountered in clinical practice. Although the majority of these tumors are benign, it has been reported that about 30% of oncology referral cases are malignant[1]. The most common malignant tumors include melanoma, squamous cell carcinoma, and lymphoma, with these malignancies being more prevalent in Caucasian patients, whereas epithelial tumors are more commonly observed in males[2]. Conjunctival malignant tumors not only have the potential to cause vision loss but can also be life-threatening in cases of metastasis[3]. The prognosis of conjunctival malignant tumors is influenced multiple factors, including age, sex, tumor staging, among others,as reviewed by several studies[4-8]. However, there are few practical prognostic models for clinical quantitative assessment of patient prognosis based on histology or staging. Therefore, in this study, we evaluated the epidemiological factors of primary conjunctival malignant tumors diagnosed between 2000 to 2019 using the SEER database. This analysis encompassed age distribution, survival rate, and risk factors related to survival time. By utilizing this data, we were able to establish a novel survival prediction model for patients with primary conjunctival malignant tumors.METHODS
data collection
The data were collected from SEER database, which represents more than 30% of American population. Our objective was to identify the independent risk factors for prognosis and subsequently construct a survival nomogram for patients with conjunctival malignant tumors based on clinicopathological data. The SEER database was used to gather all cases diagnosed with conjunctival malignant tumors according to the ICD-O-3 classification.All the patients diagnosed with conjunctival-originated malignant tumors, including melanoma, lymphoma, and carcinoma, from 2000 to 2019 were included. The inclusion criteria comprised patients with a clear diagnosis of primary conjunctival malignancies and complete clinical, pathological, and follow-up data. The exclusion criteria encompassed patients with an unclear primary tumor site, incomplete follow-up procedures, a history of other tumors, or confirmed death due to other diseases. Cases meeting the inclusion criteria were randomly assigned to either training cohorts or validation cohorts. The following information were collected for each patient: age, race, sex, tumor site (lateral), histology, stage, treatment, and other sociological factors. Histology was categorized as squamous cell carcinoma, lymphoma, malignant melanoma and others. Stage was classified as localized, regional, distant,or unknown, based on the AJCC 7th edition. The study endpoints were set at one-, three-, and five-year overall survival (OS) rates.
Statistical analysis
Patient survival time is defined as the interval from diagnosis to the last follow-up or death. Statistical significance is defined as P < 0.05 in two-tailed analysis. Utilizing X-tile software, the optimal cut-off values for age grouping were determined to be 58 and 72. The Kaplan-Meier method was used to estimate survival curves, and survival differences were assessed using the log-rank test. The Cox regression model, with hazard ratios (HRs) and 95% confidence intervals (CIs), was applied to the training cohort to identify prognostic variables.Variables selected from univariate regression were subsequently included in a forward stepwise multivariate regression analysis. Identification and calibration metrics were used to validate the nomogram model. The concordance index (C index) was calculated to assess the predictive power of the model by quantifying the discordane between observed and predicted outcomes. Receiver operating characteristic (ROC) curves and calibration curves were plotted to evaluate the model’s discrimination and calibration, respectively. The area under curve (AUC) was measured for both the training and validation sets. The calibration between predicted and actual survival rates was visualized using a calibration plot. Furthermore, decision curve analysis (DCA) was conducted to assess the clinical utility and net benefit of the prediction model. Statistical analyses were performed using IBM SPSS Statistics 23 and R software (version 4.0.3).
RESULTS
data collection
A total of 2728 cases of conjunctival malignant tumors were recorded in SEER database from 2000 to 2019, of which 1824 cases met the inclusion criteria (1216 in the development group and 608 in the validation group).The baseline characteristics of the study population, along with detailed information on survival times and causes of death, are presented in Table 1. Approximately 57.9% of the population were male. The age at onset ranged from 5 to 98 years, with a median age of 61 years. Specifically, 989 cases (54.2%) were aged under 59, 527 cases (28.9%) were aged 59 to 72 years, and 308 cases (16.9%) were aged over 72 years. The majority of the population (81.4%) were Caucasian, and 79% of the cases had localized lesions. The pathological types were categorized into four groups: squamous cell carcinoma (626 cases, 34.3%), lymphoma (557 cases, 30.5%), malignant melanoma (368 cases, 20.2%), and other types of malignant tumors (273 cases, 15.0%). The overall survival rate was 77.8% (1,419 cases). Surgery was performed on 64% (1,168 cases) of patients, while radiation therapy was administered to 1,300 cases (71.3%). Only 211 cases (11.6%) received definitive local or systemic chemotherapy.
Table 1 Baseline characteristics of patients included
|
|
|
|
|
NO. |
n percentage/%
|
|
|
Overall |
1 824 |
|
|
|||
|
Sex |
|
|
|
|||
|
|
Male |
|
1 057 |
57.9 |
|
|
|
|
Female |
|
767 |
42.1 |
|
|
|
Age |
|
|
|
|||
|
|
< 59 years |
|
989 |
54.2 |
|
|
|
|
59 - 72 years |
|
527 |
28.9 |
|
|
|
|
> 72 years |
|
308 |
16.9 |
|
|
|
Race |
|
|
|
|||
|
|
Caucasian |
|
1 484 |
81.4 |
|
|
|
|
African-american |
|
90 |
4.9 |
|
|
|
|
Other |
|
250 |
13.7 |
|
|
|
Lateral |
|
|
|
|
||
|
|
Left |
|
|
879 |
48.2 |
|
|
|
Right |
|
|
863 |
47.3 |
|
|
|
Bilateral |
|
|
82 |
4.5 |
|
|
Histology |
|
|
|
|||
|
|
Squamous cell carcinoma |
|
626 |
34.3 |
|
|
|
|
Lymphoma |
|
557 |
30.5 |
|
|
|
|
Malignant melanoma |
|
368 |
20.2 |
|
|
|
|
Other |
|
273 |
15.0 |
|
|
|
Stage |
|
|
|
|||
|
|
Localized |
|
1 441 |
79.0 |
|
|
|
|
Regional |
|
129 |
7.1 |
|
|
|
|
Distant |
|
67 |
3.7 |
|
|
|
|
Unknown |
|
187 |
10.3 |
|
|
|
Surgery |
|
|
|
|||
|
|
No/Unknown |
|
656 |
36.0 |
|
|
|
|
Yes |
|
1 168 |
64.0 |
|
|
|
Radiation |
|
|
|
|||
|
|
No/Unknown |
|
524 |
28.7 |
|
|
|
|
Yes |
|
1 300 |
71.3 |
|
|
|
Chemotherapy |
|
|
|
|||
|
|
No/Unknown |
|
1 613 |
88.4 |
|
|
|
|
Yes |
|
211 |
11.6 |
|
|
|
Marital |
|
|
|
|||
|
|
No/Unknown |
|
932 |
51.1 |
|
|
|
|
Yes |
|
892 |
48.9 |
|
|
|
Income |
|
|
|
|||
|
|
< 5500$ |
|
427 |
23.4 |
|
|
|
|
> 5500$ |
|
1 397 |
76.6 |
|
|
|
Rural-Urban |
|
|
|
|||
|
|
Rural |
|
1 622 |
88.9 |
|
|
|
|
Urban |
|
202 |
11.1 |
|
|
|
Status |
|
|
|
|||
|
|
Alive |
|
1 419 |
77.8 |
|
|
|
|
Dead |
|
405 |
22.2 |
|
|
Univariate and multivariate Cox regression analysis
The results of univariate and multivariate Cox analyses for OS are presented in Table 2. Variables that were statistically significant in the univariate Cox regression (P < 0.05) were included in the multivariate analysis, namely age, histology, stage, marital status, and income.Table 2 Univariate and multivariate Cox analyses of patients included
|
Variable |
Univariate Analysis |
|
Multivariate Analysis |
|||||
|
HR |
95% CI |
P value |
|
HR |
95% CI |
P value |
||
|
Sex |
|
|
0.225 |
|
|
|
|
|
|
|
Male |
1(ref) |
|
|
|
|
|
|
|
|
Female |
0.885 |
0.725-1.079 |
0.225 |
|
|
|
|
|
Age |
|
|
0.017 |
|
|
|
<0.001 |
|
|
|
< 59 years |
1(ref) |
|
<0.001 |
|
1(ref) |
|
|
|
|
59 - 72 years |
5.244 |
3.979-6.913 |
<0.001 |
|
5.095 |
3.860-6.726 |
<0.001 |
|
|
> 72 years |
15.380 |
11.680-20.253 |
<0.001 |
|
13.783 |
10.425-18.223 |
<0.001 |
|
Race |
|
|
0.020 |
|
|
|
|
|
|
|
White |
1(ref) |
|
<0.001 |
|
|
|
|
|
|
Black |
0.404 |
0.215-0.756 |
0.005 |
|
|
|
|
|
|
Other |
0.404 |
0.271-0.601 |
<0.001 |
|
|
|
|
|
Lateral |
|
|
0.006 |
|
|
|
|
|
|
|
Left |
1(ref) |
|
0.010 |
|
|
|
|
|
|
Right |
1.054 |
0.866-1.283 |
|
|
|
|
|
|
|
Bilateral |
0.329 |
0.155-0.700 |
0.030 |
|
|
|
|
|
Histology |
|
|
<0.001 |
|
|
|
<0.001 |
|
|
|
Squamous cell carcinoma |
1(ref) |
|
<0.001 |
|
1(ref) |
|
|
|
|
Lymphoma |
0.340 |
0.256-0.452 |
<0.001 |
|
0.456 |
0.342-0.610 |
<0.001 |
|
|
Malignant melanoma |
0.873 |
0.682-1.118 |
0.283 |
|
0.954 |
0.737-1.234 |
0.720 |
|
|
Other |
0.701 |
0.529-0.930 |
0.014 |
|
0.631 |
0.468-0.851 |
0.003 |
|
Stage |
|
|
0.038 |
|
|
|
0.010 |
|
|
|
Localized |
1(ref) |
|
0.040 |
|
1(ref) |
|
|
|
|
Regional |
1.374 |
0.973-1.942 |
0.071 |
|
1.031 |
0.720-1.477 |
0.867 |
|
|
Distant |
1.667 |
1.090-2.550 |
0.018 |
|
2.345 |
1.491-3.688 |
<0.001 |
|
|
Unknown |
1.145 |
0.827-1.587 |
0.415 |
|
0.999 |
0.719-1.380 |
0.997 |
|
Surgery |
|
|
0.025 |
|
|
|
|
|
|
|
No/Unknown |
1(ref) |
|
|
|
|
|
|
|
|
Yes |
1.268 |
1.027-1.565 |
0.027 |
|
|
|
|
|
Radiation |
|
|
<0.001 |
|
|
|
|
|
|
|
No/Unknown |
1(ref) |
|
|
|
|
|
|
|
|
Yes |
0.561 |
0.440-0.715 |
<0.001 |
|
|
|
|
|
Chemotherapy |
|
|
0.795 |
|
|
|
|
|
|
|
No/Unknown |
1(ref) |
|
|
|
|
|
|
|
|
Yes |
1.040 |
0.774-1.399 |
0.794 |
|
|
|
|
|
Marital |
|
|
<0.001 |
|
|
|
0.026 |
|
|
|
No/Unknown |
1(ref) |
|
|
|
1(ref) |
|
|
|
|
Yes |
0.702 |
0.577-0.854 |
<0.001 |
|
0.797 |
0.652-0.973 |
|
|
Income |
|
0.001 |
|
|
|
0.025 |
||
|
|
< 5500$ |
1(ref) |
|
|
|
1(ref) |
|
|
|
|
> 5500$ |
0.692 |
0.558-0.857 |
0.001 |
|
0.781 |
0.629-0.970 |
|
|
Rural-Urban |
|
|
0.002 |
|
|
|
|
|
|
|
Rural |
1(ref) |
|
|
|
|
|
|
|
|
Urban |
0.642 |
0.490-0.840 |
0.001 |
|
|
|
|
construction and validation of prognostic nomogram
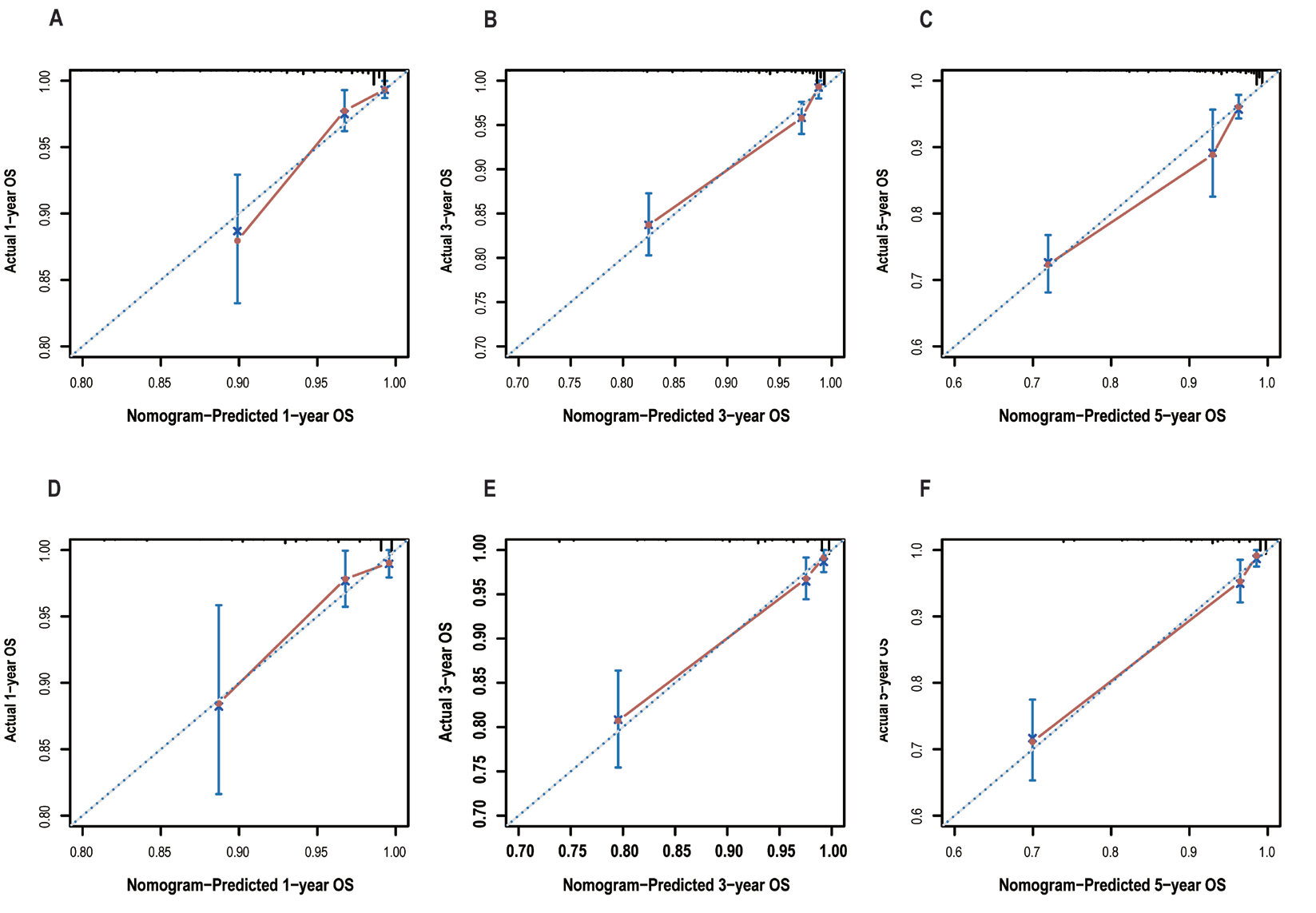
Five independent factors influencing overall survival were utilized to construct a prognostic nomogram, as illustrated in Figure 2. The C-index of the constructed model was 0.79, indicating a convincing consistency between the predicted and observed survival outcomes at 1-, 3-, and 5-years.Figure 5 displays the calibration plots of nomogram, which demonstrate that the predicted overall survival rate from model closely agree with actual overall survival rate at 1-, 3-, and 5-years, for both training and validation group.
Figure 1 OS for the patients with primary conjunctival malignant tumors using Kaplan–Meier analysis and log-rank test
(A) 1-year net benefit in training cohort; (B) 3-year net benefit in training cohort; (C) 5-year net benefit in training cohort; (D) 1-year net benefit in validation cohort; (E) 3-year net benefit in validation cohort. (F) 5-year net benefit in validation cohort. According to multivariate analyses, the survival outcomes were better in those patients with lymphoma, younger age, and localized lesions. Patients with squamous cell carcinoma or malignant melanoma had shorter survival time than those with lymphoma or others.
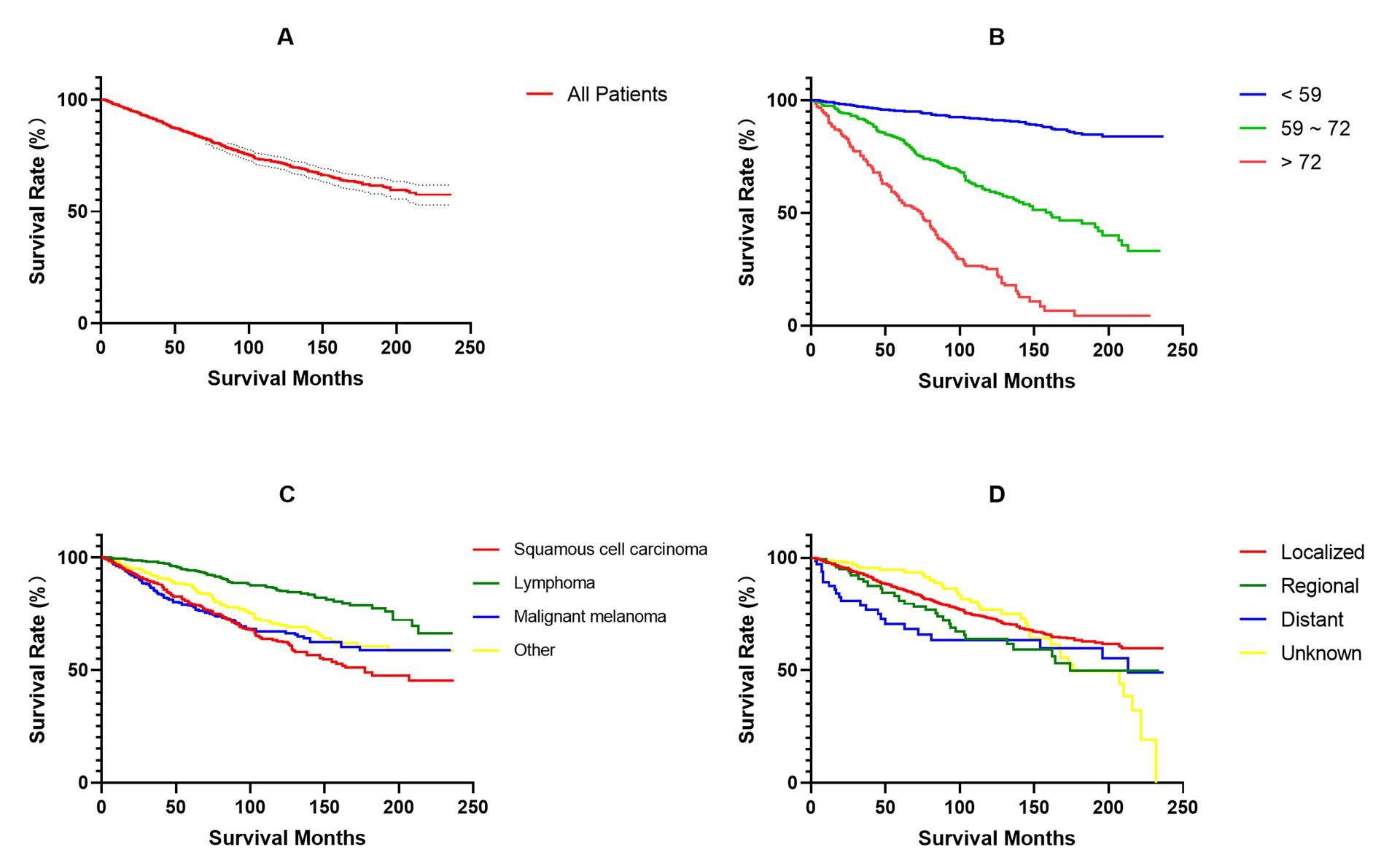
Figure 2 Nomogram predicting 1-, 3-, and 5-year OS for patients with primary conjunctival malignant tumors
The C-index of the constructed model was 0.79, which represents the convincing consistency between the predicted and observed survival outcomes at 1-, 3-, and 5- years.
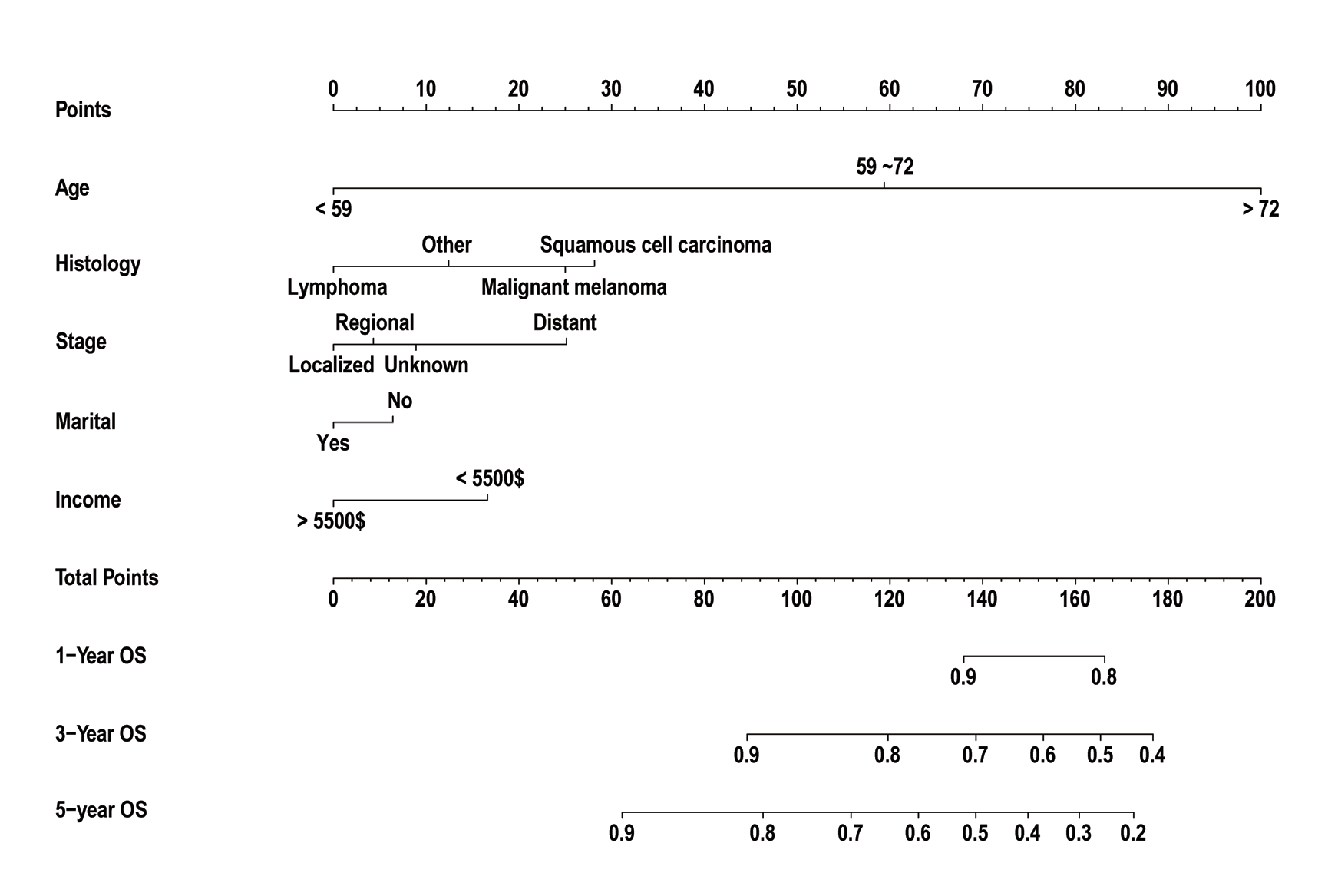
Figure 3 Receiver operating characteristic curve analysis for evaluating the accuracy of the 1-, 3-, and 5-year nomogram
The death rate was expressed as a continuous variable. The AUC values for 1-, 3-, and 5-year predicted mortality were 0.824, 0.796, and 0.815 in the training set; 0.750, 0.820 and 0.838 in the validation set, respectively
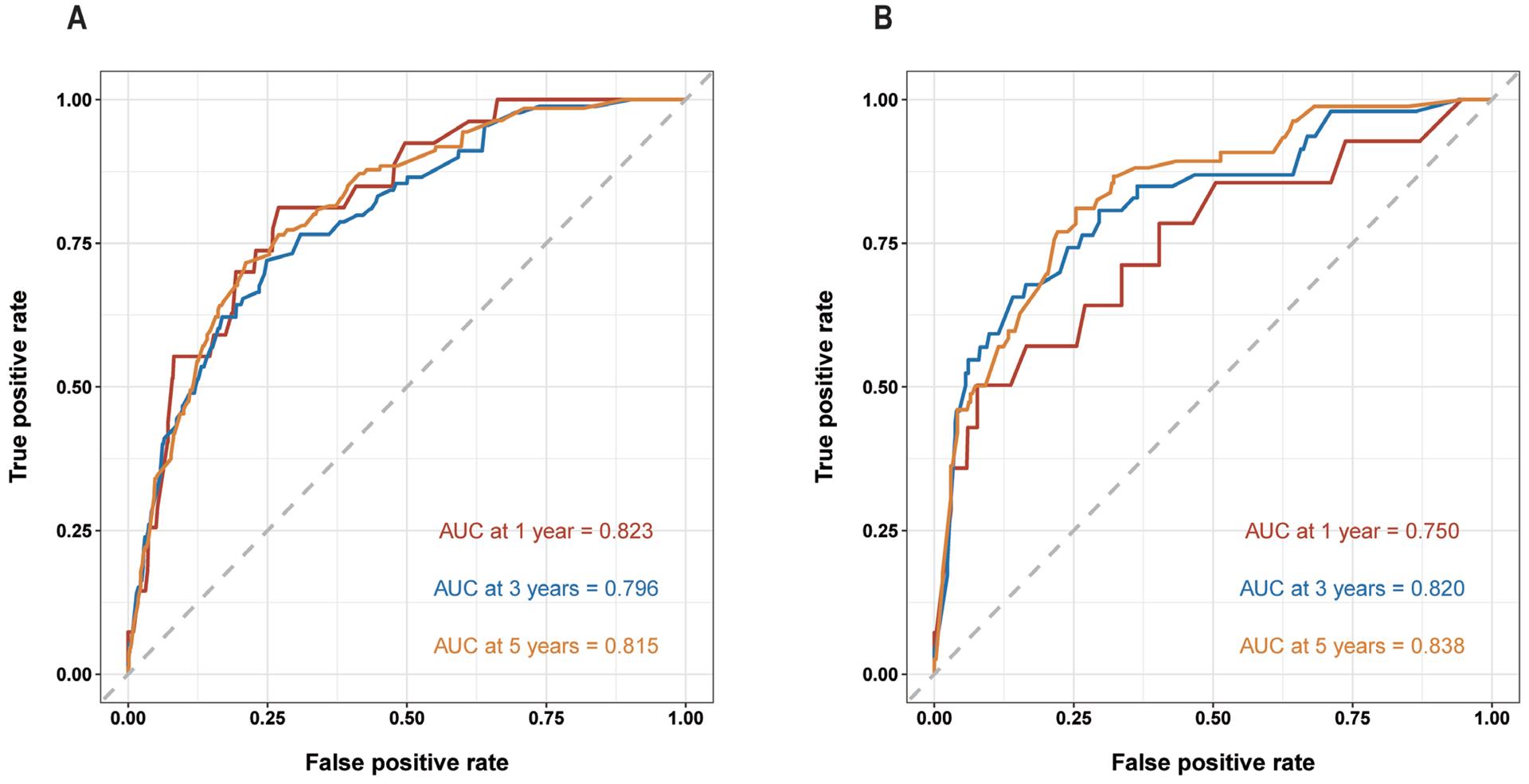
Figure 4 Decision curve analysis to assess the clinical utility and net benefit of the prediction model
(A) 1-year net benefit in training cohort; (B) 3-year net benefit in training cohort; (C) 5-year net benefit in training cohort; (D) 1-year net benefit in validation cohort; (E) 3-year net benefit in validation cohort. (F) 5-year net benefit in validation cohort. The decision curve analyses results indicate the model provides prominent benefits in prognosis prediction at 1-, 3-, and 5- years to patients with primary conjunctival malignant tumor.
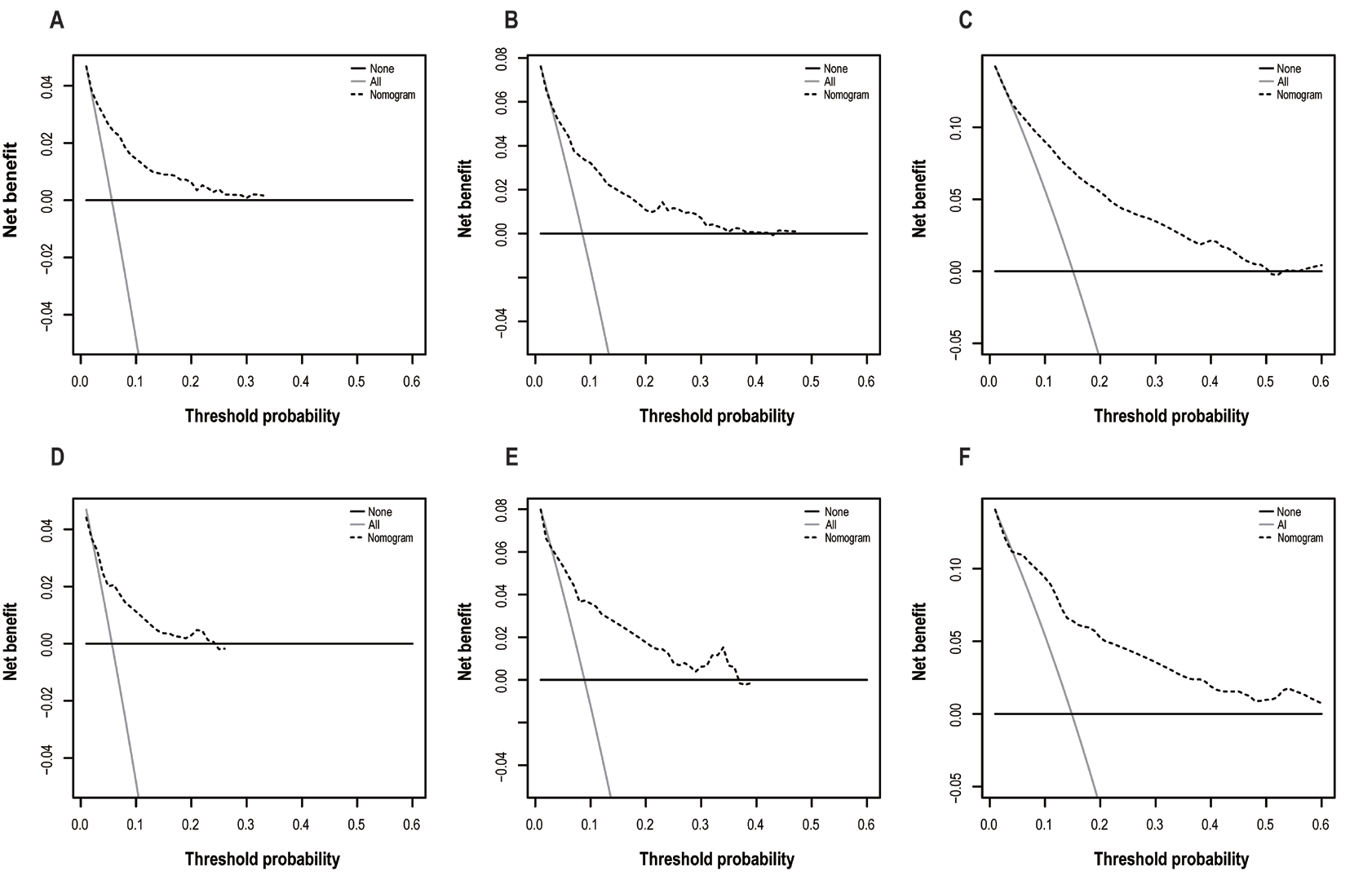
Figure 5 Calibration plots of the nomogram for predicating 1-, 3-.and 5-years OS
Nomogram predicted OS is plotted on the x-axis, actual OS is plotted on the y-axis. Training group for 1-year (A), 3-year (B) and 5-year (C). Validation group for 1-year (D), 3-year (E) and 5-year (F).
DISCUSSION
Malignant tumors of the conjunctiva are uncommon and can arise from squamous epithelium, melanocytes, and lymphocytic infiltrates in the conjunctival stroma. These tumors are categorized into three distinct types, each with unique pathological features, mechanisms of development, and prognoses. In this study, two utilized data from the National Cancer Institute's SEER (Surveillance, Epidemiology, and End Results) program, which is a highly authoritative source of information on malignant tumor incidence in the United States and has been extensively used in epidemiological research on conjunctival tumors. Nomograms are now widely used to predict prognosis, and a useful nomogram with good accuracy and clear prognostic indication can aid in making better clinical decisions. In the present study, we developed a prognostic model based on large sample of data to predict the prognosis of malignant conjunctival tumors, which are rare but life-threatening.To our knowledge, no pre-existing prognosis prediction nomogram based on the histology and stage had been developed for malignant conjunctival tumors. Wang reported a predicted prognosis model using SEER database; however, their model emphasized sociological factors such as marital status without considering the tumor’s histology, which is important[9]. The c-index in their nomogram was 0.747. In our nomogram, we included tumor size, site (lateral), histology, stage and treatment to identify significant variables. As a result, our c-index was 0.79, which is higher than the previous report, indicating a more accuracy model. In terms of predicting the 1-, 3- and 5-year overall survival rate, the AUC were 0.824, 0.796, and 0.815 in the training set, and 0.750, 0.820 and 0.838 in the validation set in our nomogram. In the previous report, the AUC values for 3- and 5-year were 0.715 and 0.74166 in the standard mode, and 0.697 and 0.75249 in verification mode, respectively [9]. The comparison also shown that our nomogram may have a better predictive effect than previous report.
Age is a significant risk factor for many diseases, especially cancer. The current global trend towards an aging population has led to changes in the age distribution, cancer spectrum, and mortality rate among cancer patients. Previous studies have demonstrated a correlation between the incidence of conjunctival malignant tumors and patient age[10]. We observed an increased incidence of these tumors with advancing age, particularly in individuals over 50 years old. Younger patients generally have a better prognosis, which is supported by our research findings. On the other hand, elderly patients often have multiple systemic comorbidities, such as hypertension, diabetes, or coronary artery disease, which may be associated with poorer outcomes.
It is well-known that biological behavior of tumors differ according to histology. Our study revealed that patients with lymphoma had relatively longer survival time, while patients with melanoma or squamous cell carcinoma tended to have worse prognosis. Relevant studies have shown that lymphoma-related survival rates depend on the subtype, ranging from 97% (ENMZL) to 9% (MCL)[11]. Research has shown that male gender, T4 and N1 staging are also significant prognostic factors for melanoma [10] . Predisposing conditions for lymphoma include benign reactive lymphoid hyperplasia, immunodeficiency (HIV), immunosuppression, and chronic inflammation/infection (Helicobacter pylori, Chlamydia psittaci). For squamous cell carcinoma (SCC), a higher incidence is associated with chronic sun exposure, immunodeficiency (HIV), organ transplantation, autoimmune diseases, actinic keratosis, and prolonged exposure to cigarette smoke[12]. As for melanoma, predisposing conditions include primary acquired melanosis (PAM), chronic nevi, and chronic sun exposure[13].
Several studies have reported that race may play a role in the incidence of malignant conjunctival tumors, as black individuals are more likely to have melanoma [14,15]. Yu et al. previously found that the incidence of conjunctival malignant tumors in white males increased by 295% over a 27-year period from 1973 to 1999 [16]. In the United States, conjunctival squamous cell carcinoma (SCC) is rare, with an incidence rate of 0.03 per 100,000 people, and the incidence is 5 times higher in males and whites[12]. However, the spectrum of ocular squamous lesions is different in Africa, where it is almost equally common in females and males and occurs at a younger age than in the United States[17]. In our study, race was not an independent prognostic factor.
Similar to race, sex may be associated with incidence of conjunctival tumors, but not with prognosis[18]. Diao's regression analysis results indicated a higher risk of malignant conjunctival tumors in males[9]. Another study showed that ocular squamous tumors were more common in males, with a male-to-female ratio of 3.3:1.0 [19].This is consistent with our research. However, other analyses suggested that gender was not associated with the incidence or prognosis of conjunctival tumors [20]. Currently, there is no clear explanation for why males have a higher risk of developing such tumors, but possible related risk factors may include exposure to ultraviolet radiation, outdoor activities, and hormonal factors, depending on study populations or analyzed tumor types.
Surgical resection is the primary treatment method for these tumors. However, in the past 20 years, the treatment approach for SCC has undergone changes, and local therapies such as mitomycin C, 5-fluorouracil, and interferon α-2 have been used as primary and adjuvant treatments. Both conjunctival squamous cell carcinoma and melanoma have relatively high rates of local recurrence after treatment, ranging from 30-41% [21]and 50%-70% [22,23], respectively. But melanoma has a higher tendency for metastasis. Multivariate analysis of all malignant conjunctival tumors indicates that the age at diagnosis, T stage, and N stage at presentation are important determinants of overall survival (OS)[10]. In general, the prognosis of conjunctival tumors depends on multiple factors, including tumor type, malignancy, early diagnosis, timely treatment, and the patients' overall health condition. Tumors detected early and treated promptly usually have a better prognosis, while larger, highly malignant tumors discovered at a later stage may have a less favorable prognosis[24].
Several studies, including current study, have demonstrated that marital status or economic income are risk factors for both incidence and prognosis of conjunctival cancer, as social relationship and disposable income may impact patient’s psychological status, nursing care and treatment choice. In some studies, it has been found that being male, single, divorced, and widowed are risk factors associated with the incidence of primary conjunctival cancer[9]. Some studies have also indicated that unmarried individuals are more likely to experience metastatic disease, inadequate treatment, and cancer-related mortality[25]. However, marital status has not been found to be a significant survival predictor for conjunctival squamous cell carcinoma (SCC)[10].
In this study, we conducted a multifactor analysis and found that patients with lymphoma, younger age, and localized lesions had better survival outcomes. In contrast, patients with squamous cell carcinoma or malignant melanoma had shorter survival times compared to lymphoma or other types of patients. We developed a survival outcome using a nomogram model, which comprehensively considered multiple factors and provided more comprehensive and personalized treatment recommendations for doctors and patients. This model is based on various predictive indicators and accurately predicts patients' prognosis. We also used the area under the curve (AUC) value to evaluate the model's performance, which is an important index for measuring predictive model accuracy. In our research, we obtained satisfactory AUC values, indicating that our constructed nomogram model exhibits a high level of accuracy in predicting the prognosis of conjunctival tumor patients. These findings provide powerful decision-making support for clinicians to devise more effective treatment plans and improve the treatment outcomes and prognosis of conjunctival malignant tumor patients.
However, some limitations in our study should be mentioned. Firstly, the SEER database has certain limitations, such as restricted regional coverage and incomplete or missing treatment information, along with the absence of important data like genetic information. These gaps may introduce potential bias in sample selection and result in less accurate analysis of confounding factors. Second, we acknowledge that the three types of tumors included in this study—melanoma, squamous cell carcinoma, and lymphoma—exhibit significant biological differences, particularly in terms of recurrence, metastasis, and prognosis. These differences may limit the comparability of these tumors and affect the clinical relevance of combining them into a single predictive model. However, due to the relative rarity of conjunctival malignant tumors, as well as relatively lower attention in the field of medical research, constructing separate models for each tumor type is often constrained by the small sample size available. Furthermore, there is limited research or widespread application of predictive models and risk assessment tools for malignant conjunctival tumors currently. Therefore, the aim of our study was to develop a broadly applicable prognostic tool that could assist clinicians in quickly assessing survival outcomes for patients with different types of conjunctival malignancies. While we recognize the limitations of a generalized model, we believe it provides valuable insights for clinical practice, especially in cases where specific tumor diagnosis may not be immediately clear. We agree that future studies should focus on developing individual predictive models for each tumor type, while conducting cross-analyses that integrate genetic and clinical data from additional databases. This approach aims to enhance the accuracy and clinical relevance of prognostic predictions.
CONCLUSION
In this study, we established that age, tumor histology and tumor stage are independent prognostic factors for conjunctival malignant tumors. Furthermore,an efficient and accurate survival prediction model was developed. Additional validation might be needed in the future study.Correction notice
NoneAcknowledgements
NoneAuthor Contributions
(I) Conception and design: Jianjun Gu(II) Administrative support: Jianjun Gu
(III) Provision of study materials or patients: Miao Chen
(IV) Collection and assembly of data: Lixia Lin
(V) Data analysis and interpretation: Lixia Lin
(VI) Manuscript writing: All authors
(VII) Final approval of manuscript: All authors