Nanophthalmos associated with acute ischemic optic neuropathy: a case report and literature review
关键词
摘要
Nanophthalmos, also referred to as true microphthalmos, is a rare congenital ocular developmental disorder characterized by a significantly reduced globe volume compared to age-matched normative parameters, often accompanied by multiple ocular comorbidities such as glaucoma, uveal effusion syndrome, and retinopathy, as well as potential systemic anomalies. Acute ischemic optic neuropathy (AION) is an acute-onset optic nerve disorder caused by interruption or substantial reduction of blood perfusion to the optic nerve head, which typically leads to rapid visual deterioration, ranging from moderate vision loss to complete blindness if not promptly managed. Clinically, the co-occurrence of nanophthalmos and AION is extremely rare, with limited literature reporting this comorbidity, making it prone to misdiagnosis as optic neuritis due to overlapping manifestations including acute visual decline and optic disc edema.We herein report a case of a 40-year-old male patient with bilateral nanophthalmos complicated by non-arteritic anterior ischemic optic neuropathy (NA-AION) in the right eye and optic atrophy in the left eye. The patient presented with a 5-day history of progressive right ocular pain and visual loss, with best-corrected visual acuity (BCVA) of counting fingers at 50 cm in the right eye and 20/1000 in the left eye at initial presentation. Ocular examinations confirmed key features of nanophthalmos, including shortened axial lengths (16.48 mm in the right eye and 18.49 mm in the left eye) and diffuse scleral thickening, along with typical AION findings such as optic disc edema, peripapillary hypoperfusion on fundus fluorescein angiography, and global visual field defects.
Conservative treatment was initiated, including intravenous methylprednisolone (adjusted from 250 mg to 80 mg due to transient visual deterioration with high doses), subcutaneous anisodamine for microcirculation improvement, and neurotrophic support. At the 1-month follow-up, partial vision recovery was achieved, with the right eye’s BCVA improving to 20/1000 and significant resolution of optic disc edema confirmed by serial optical coherence tomography. The underlying pathophysiological mechanism is hypothesized to involve anatomical abnormalities of nanophthalmos, such as shortened axial length, thickened sclera, and increased choroidal thickness, which may compress vortex veins, disrupt posterior ciliary artery perfusion, and induce optic nerve head hypoperfusion. This article details the clinical characteristics, diagnostic process, treatment response, and potential pathogenic mechanisms of this rare comorbidity, aiming to provide clinical insights for the early diagnosis and management of similar cases and highlight the importance of distinguishing AION from optic neuritis in patients with nanophthalmos.
全文
HIGHLIGHTS
1.Critical Discoveries and Outcomes
1) Systematically reported a rare case of bilateral nanophthalmos complicated with right-eye non-arteritic anterior ischemic optic neuropathy (NA-AION) and left-eye optic atrophy, confirming that this complication can occur independently in nanophthalmic patients without a glaucoma background, revising the previous notion that such optic neuropathy requires concurrent glaucoma.
2) Revealed that anatomical abnormalities of nanophthalmos (short axial length, scleral thickening, choroidal hypertrophy) are the core pathogenic factors of AION: compressing vortex veins and disrupting posterior ciliary artery perfusion led to optic disc hypoperfusion, rather than direct high intraocular pressure effects, providing novel evidence for the disease’s pathological mechanism.
3) Verified the efficacy of conservative treatment (low-dose glucocorticoids + microcirculation improvement + neurotrophic support) for this complication: the patient’s right-eye best-corrected visual acuity (BCVA) improved from counting fingers at 50 cm to 20/1000, with significant resolution of optic disc edema, breaking the traditional model of surgical dependence in some cases.
4) Clarified key differential points between nanophthalmos-associated AION and optic neuritis: absence of optic nerve enhancement on MRI, and segmental optic disc hypoperfusion on fluorescein fundus angiography (FFA), effectively avoiding clinical misdiagnosis.
2.Methodological Innovations
Adopted a multimodal imaging combined diagnostic strategy: integrated spectral-domain optical coherence tomography (OCT), FFA, B-mode ultrasound, ultrasound biomicroscopy (UBM), visual evoked potential (VEP), and orbital MRI to comprehensively quantify ocular anatomical abnormalities (axial length, scleral thickness, choroidal thickness) and pathological changes (optic nerve edema, retinal nerve fiber layer thinning), improving the accuracy of rare disease diagnosis.
3.Prospective Applications and Future Directions
1) Clinical application: Provides a new target for follow-up management of nanophthalmic patients; recommends incorporating optic nerve perfusion assessment (e.g., OCT angiography [OCTA]) into routine examinations, and prioritizing AION screening in patients with sudden visual loss and optic disc edema to avoid misdiagnosis as optic neuritis.
2) Therapeutic expansion: Offers an evidence-based basis for the application of conservative treatment in such diseases; future research may further explore combined regimens of microcirculation-improving drugs and neurotrophic agents to optimize non-surgical treatment efficacy.
3) Research directions: Large-sample, multi-center studies are needed to verify the incidence and risk factors of nanophthalmos-associated AION; clarify the molecular correlation between anatomical abnormalities and vascular dysfunction via gene sequencing; explore the application of surgical interventions (e.g., scleral windowing) in refractory cases to improve clinical guidelines.
Nanophthalmos (also known as true microphthalmos) is an uncommon congenital ocular disorder, characterized by an eye size that is significantly smaller than the norm, and it is, often accompanied by other ocular or systemic abnormalities[1]. Despite its low incidence, the clinical manifestations and complications of nanophthalmos can severely impact a patient's vision[2]. In recent years, with enhanced understanding of the anatomical and physiological characteristics , researchers have acknowledged its link to a range of ocular diseases, including glaucoma, uveitis, and retinopathy. Nevertheless, there is a scarcity of literature that delves into the co-occurrence of nanophthalmos and acute ischemic optic neuropathy (AION).
AION refers to acute damage to the optic nerve that stems from an interruption or a substantial decrease in blood supply, which can potentially result in severe vision impairment or even blindness. The pathogenesis of AION is intricate, often, associated with systemic vascular diseases, local hemodynamic changes, or anatomical abnormalities[2]. Although there has been some clinical headway in the diagnosis and treatment of AION, research on its underlying mechanisms and management approaches in patients with nanophthalmos is still rather limited.
Here, we present a case of nanophthalmos complicated by AION, emphasizing its clinical characteristics, diagnostic procedures, treatment results, and possible underlying mechanisms. This rare case provides fresh perspectives on the risk factors and management strategies for optic neuropathy in the context of nanophthalmos.
CASE REPORT
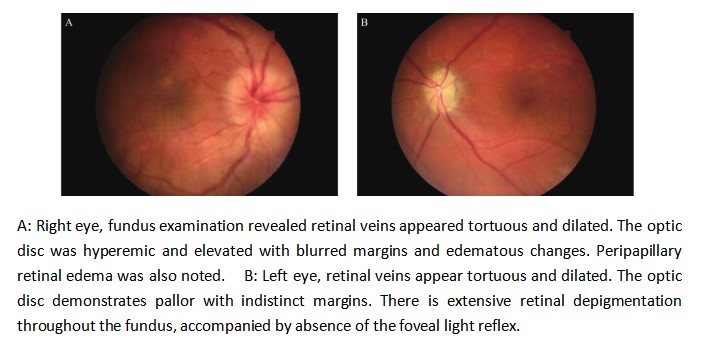
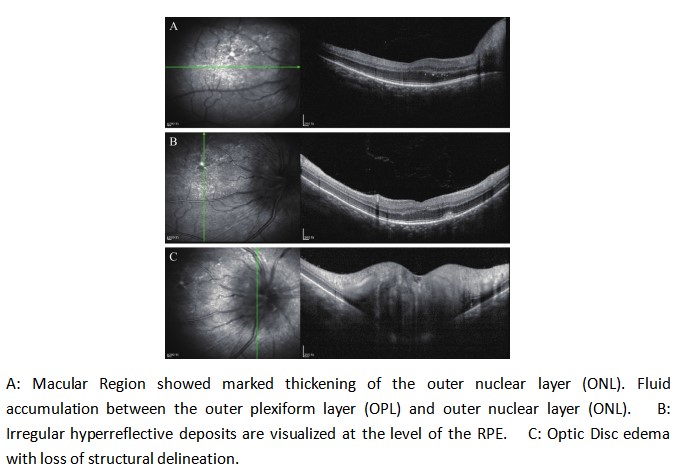
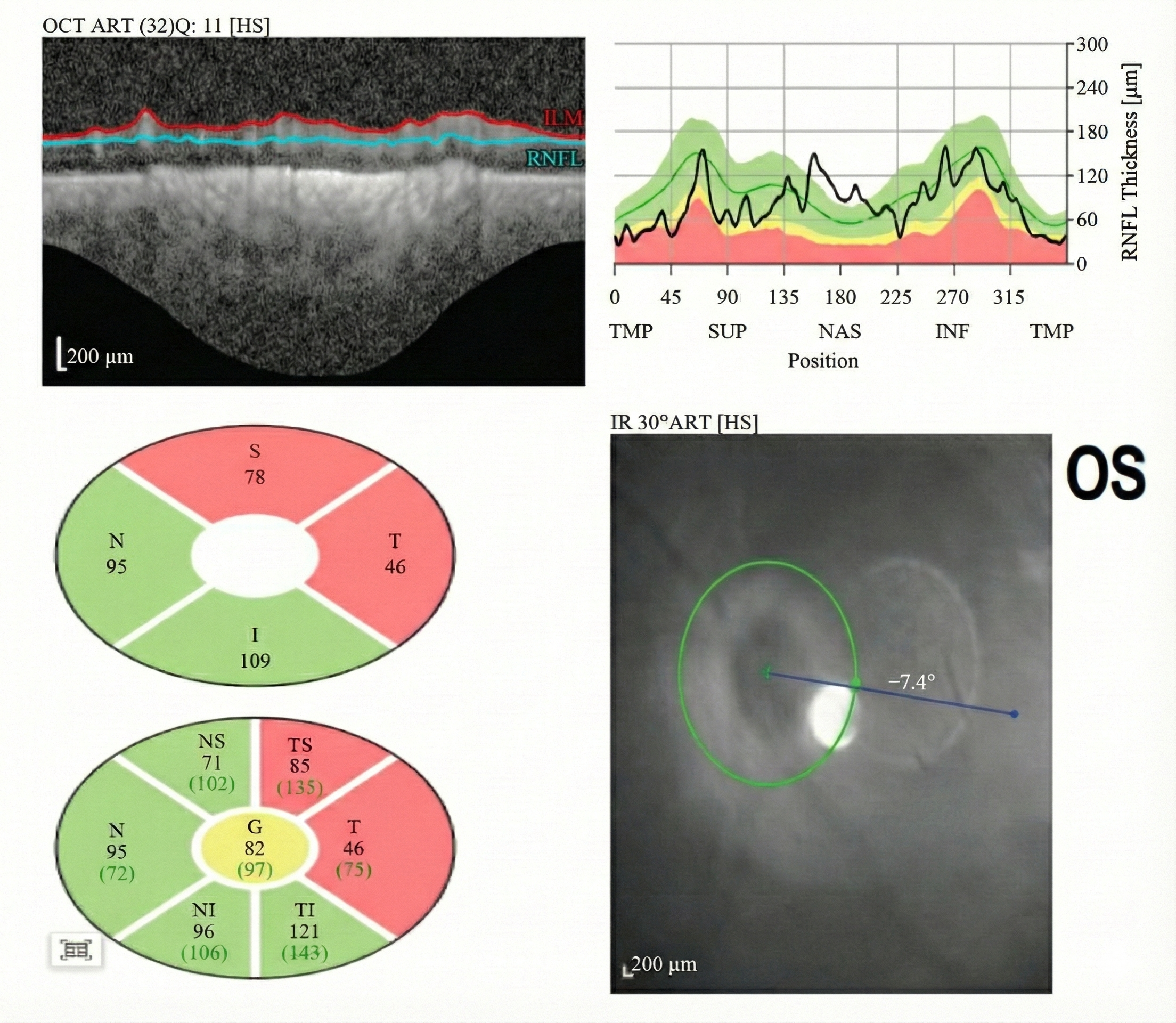
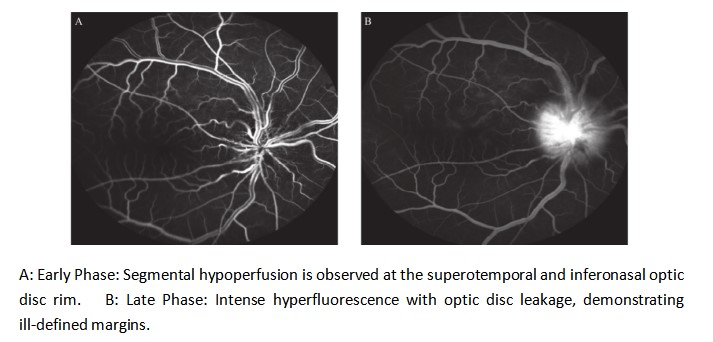
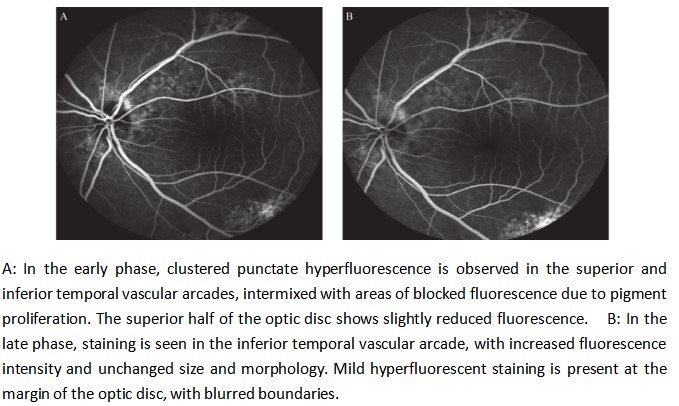
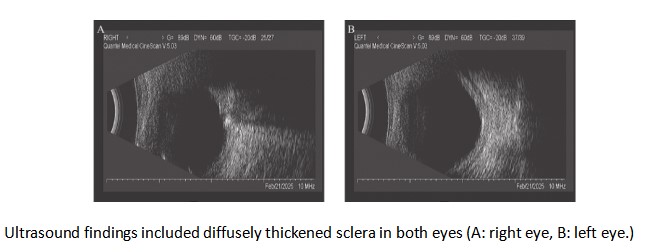
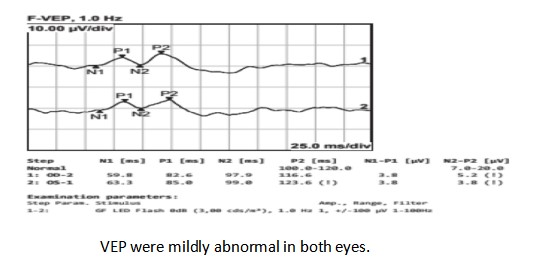
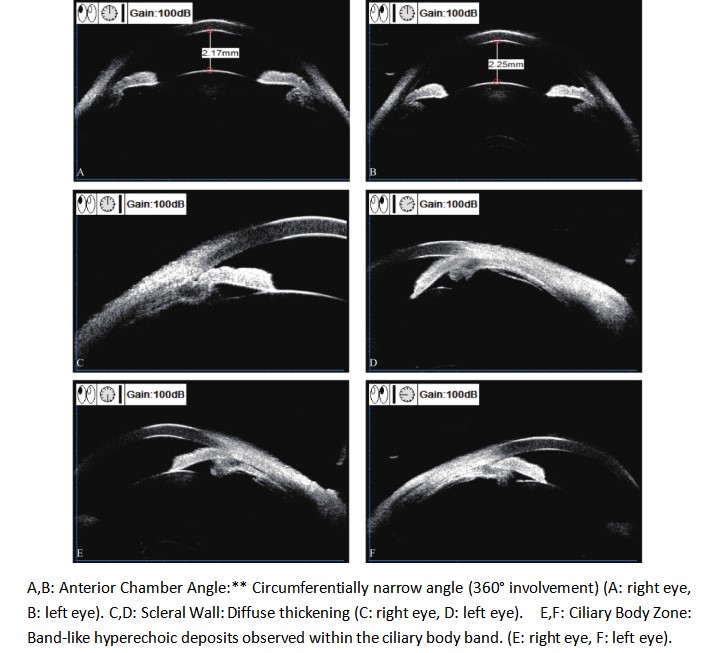
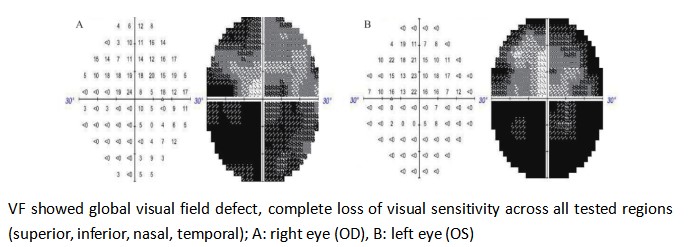
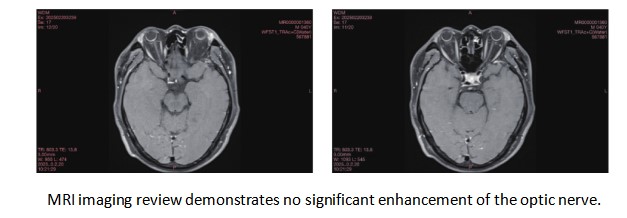
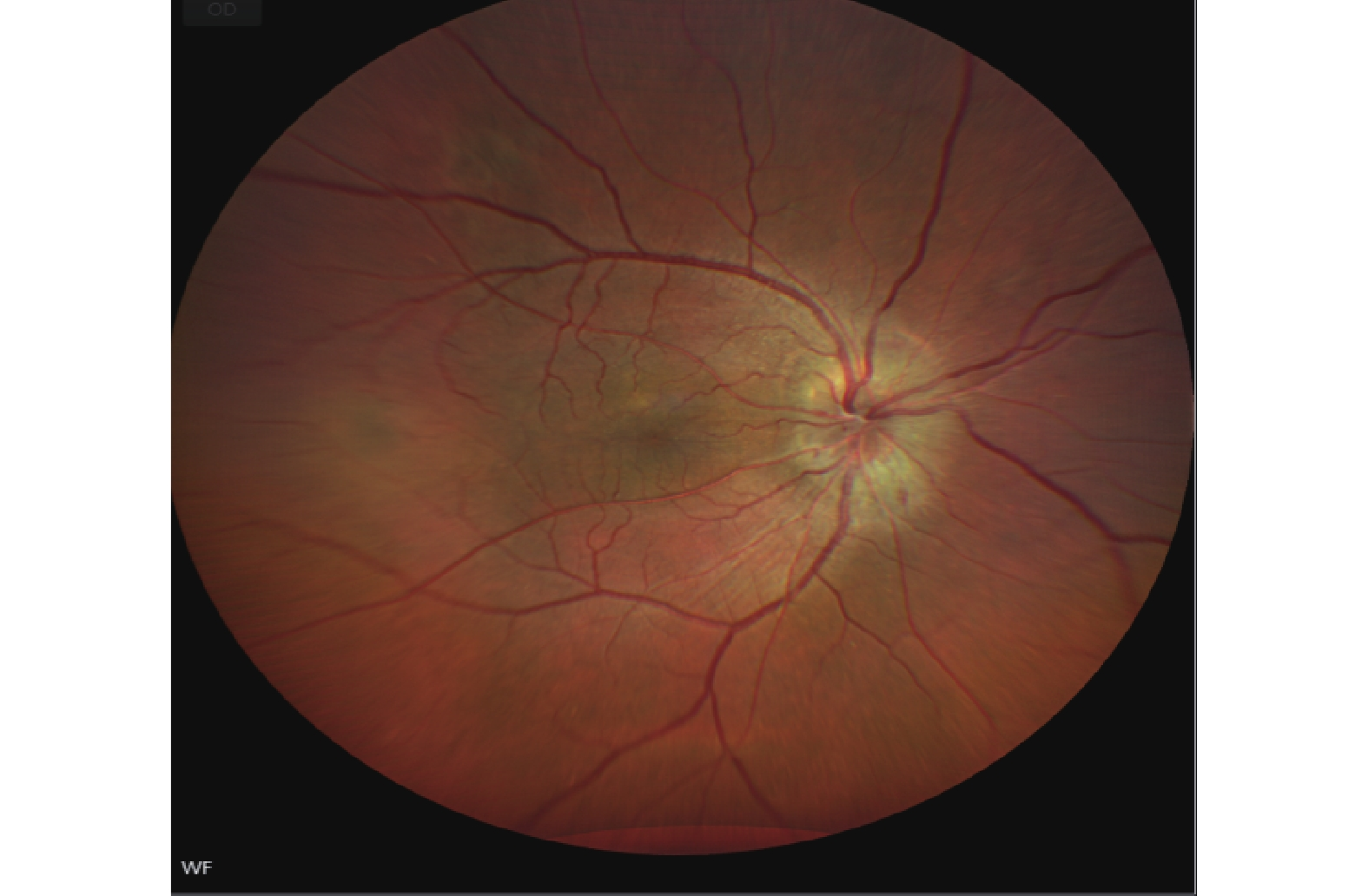
A 40-year-old male patient presented to our ophthalmology department, complaining of a 5-day history of progressively worsening right ocular pain and visual decline. Upon examination, his best-corrected visual acuity(BCVA) was counting fingers (CF) at 50 cm in the right eye (OD) and 20/1000 in the left eye (OS). The intraocular pressure measured 14 mmHg in the right eye and 13 mmHg in the left eye. The anterior chamber depth was 2.5 times the central corneal thickness (CCT) centrally and 0.25 times the CCT peripherally in both eyes. The pupils were round and equal, measuring 3.0 mm in diameter, with brisk light reflex bilaterally. The relative afferent pupillary defect (RAPD) was negative. Fundus examination of the right eye revealed optic disc edema with hyperemia. The elevated margins of the optic disc were indistinct, and there was accompanying retinal edema in the peripapillary region. The left optic disc appeared pale with blurred margins. Both eyes showed tortuous and dilated veins with an arteriovenous (A:V) ratio of 1:3. There was an absent foveal reflex, and the left eye additionally showed widespread retinal depigmentation (Figure 1A, B). Spectral-domain optical coherence tomography (SD-OCT) demonstrated three key findings in the right macula: significant thickening of the outer nuclear layer, subretinal fluid accumulation between the outer plexiform and outer nuclear layers, and irregular hyperreflective deposits at the level of the retinal pigment epithelium (RPE). Concurrent papilledema was also documented (Figure 2). In the left eye, fundus examination revealed reduced retinal nerve fiber layer (RNFL) thickness in the circumpapillary region (Figure 3) and irregular hyperreflective deposits at the RPE level within the macular area. Fundus fluorescein angiography of the right eye demonstrated early-phase hypoperfusion in the peripapillary temporal and nasal quadrants (Figure 4A), with intense late-phase dye leakage extending from the optic disc exhibiting ill-defined margins (Figure 4B). Fundus fluorescein angiography of the left eye demonstrated persistent hypoperfusion and diminished fluorescence intensity in the superior hemi-disc region throughout the late-phase angiographic sequence (Figure 5). B-scan ultrasonography revealed diffuse scleral thickening bilaterally (Figure 6). Ocular biometry documented reduced axial lengths, measuring 16.48 mm in the right eye and 18.49 mm in the left eye. Visual evoked potentials (VEFs) were mildly abnormal in both eyes (Figure 7). Ultrasound biomicroscopy (UBM) indicated narrow angles and localized shallow ciliary body detachment in both eyes (Figure 8). Visual Field Examination Findings: Both eyes showed a global visual field defect, with a complete loss of visual sensitivity across all tested regions (superior, inferior, nasal, temporal) (Figure 9). Laboratory investigations revealed that the erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels were within normal reference ranges. Orbital MRI imaging review showed no significant enhancement of the optic nerve (Figure 10).
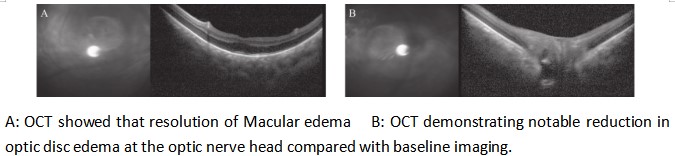
Based on a synthesis of the ophthalmic and imaging assessments, a definitive diagnosis was established, encompassing non-arteritic anterior ischemic optic neuropathy (NA-AION) right eye, bilateral nanophthalmos, and left optic atrophy. The treatment course included intravenous methylprednisolone (Solu-Medrol) at a dose of 250 mg, which alleviated the eye pain. However, when the dose was increased to 500 mg intravenously, the patient’s vision deteriorated further. The dose was then reduced to 80 mg intravenously for 7 days. Concurrently, anisodamine was administered as a subcutaneous injection (2 ml) at the temple to improve microcirculation and provide neurotrophic support. Fundus photography at discharge revealed a significant improvement in optic disc edema in the right eye compared to admission (Figure 11). At the 1-month follow-up, the BCVA in the right eye improved to 20/1000, with significant resolution of optic disc edema. Serial optical coherence tomography (OCT) confirmed a marked reduction in disc edema compared with the baseline imaging (Figure 12).
DISCUSSION
Nanophthalmos is a rare congenital eye disorder caused by early cessation of ocular growth[1,3], with recent multi-omics studies revealing that variants in genes such as MFRP, PRSS56, and MYRF disrupt scleral and vascular development, predisposing to hemodynamic complications[4]. Clinically, it typically presents bilaterally with high hyperopia and increased risk for secondary issues like angle-closure glaucoma and uveal effusion syndrome[4].
This case presents a 40-year-old man with bilateral nanophthalmos who developed acute NA-AION in the right eye, despite normal intraocular pressures, suggesting that nanophthalmos-associated anatomical changes—such as shortened axial length and scleral thickening—may independently impair optic nerve perfusion[5,6,7]. OCT angiography studies support this, showing significantly reduced vessel density at the optic nerve head in nanophthalmos[8], which, along with the patient’s rapid visual loss, indicates that structural vulnerability rather than elevated IOP is the primary trigger for NA-AION in this context[9,10].
B-scan ultrasonography shows that both eyes have a short axial length and thickened scleral walls. The choroidal thickness in patients with nanophthalmos is typically 400-800 μm, in contrast to the normal value of less than 200 μm. This thickening is prone to cause the following pathophysiological consequences:
1.Vortex vein compression: The thickened sclera compresses the vortex veins, leading to elevated choroidal venous pressure and subsequent exudation. OCT examination of the right eye in this case revealed fluid accumulation between the outer plexiform layer (OPL) and the outer nuclear layer (ONL). Both eyes showed punctate clustered transmitted fluorescence with pigment-blocked fluorescence on FFA, suggesting retinal pigment epithelium (RPE) damage (indicating that the left eye may have had a similar undetected pathological change in the past). The above examinations provide supporting evidence for this speculation. Elevated choroidal venous pressure can also cause a decrease in the perfusion pressure difference of the posterior ciliary arteries (PCAs), thereby affecting the blood perfusion of the optic nerve and resulting in AION.
2.Choroidal vasodilation: Vascular tortuosity and dilation lead to blood stasis, increasing the risk of thrombosis and enhancing susceptibility to PCA branch embolism.
3.Direct compression: The thickened choroid mechanically compresses the traversing PCAs, causing insufficient blood supply to the prelaminar optic nerve head (ONH). Meanwhile, the short axial length may cause the optic nerve to bear abnormally high mechanical pressure at the lamina cribrosa (even with normal intraocular pressure). Concomitant scleral thickening further restricts the buffering space around the optic nerve, thereby leading to thinning of the ganglion cell layer. (Supporting evidence in this case: Optic nerve OCT of the left eye shows a reduction in the thickness of the ganglion cell layer. Fundus fluorescein angiography (FFA) shows diffuse hypofluorescence throughout the left superior optic disc.)
4.Blood-retinal barrier breakdown: Elevated hydrostatic pressure disrupts the tight junctions of the retinal pigment epithelium, allowing vasogenic edema to infiltrate the peripapillary region and obstruct the axoplasmic flow of the ONH.
Therefore, in patients with nanophthalmos, changes in the hemodynamics of the optic nerve head are likely to play an important role in the pathogenesis of NA-AION [11]. Choroidal venous hypertension can cause a "steal phenomenon", leading to blood shunting from the optic disc perfusion area. This ischemic phenomenon is most pronounced when blood pressure drops at night, thereby inducing anterior ischemic optic neuropathy (AION).
In addition,even in the absence of concurrent anterior ischemic optic neuropathy (AION) or optic atrophy, visual prognosis is likely to remain suboptimal due to high hyperopia as an independent factor.
Current literature documents sparse reports of nanophthalmos complicated by NA-AION. Ahmad et al [12]. described a 59-year-old male patient who present with acute right visual loss. A comprehensive ophthalmic examination revealed bilateral nanophthalmos, along with right optic disc swelling and choroidal thickening. Following scleral window surgery, the patient experienced substantial recovery of visual acuity and complete resolution of disc edema. Other case reports have documented patients with nanophthalmos who also had coexisting uveal effusion syndrome and glaucoma. A representative case involved a 48-year-old male who manifested unilateral uveal effusion syndrome (UES) accompanied by mild optic disc swelling in the left eye. His vision significantly improved after scleral window surgery [13]. All these cases suggest that the occurrence of AION in patients with nanophthalmos may be related to choroidal thickening and a crowded optic nerve head. Patients with nanophthalmos typically have a reduced globe volume and thickened scleral walls. These anatomical features obstruct vortex venous drainage and elevate suprachoroidal pressure, thereby impairing microcirculation at the optic nerve head. This venous congestion may induce optic nerve head hypoperfusion, thus increasing the susceptibility to non-arteritic anterior ischemic optic neuropathy.
Compared with aforementioned cases, the unique aspect of our case is that the patient did not have glaucoma but suddenly developed AION in the right eye 5]. This indicates that in patients with nanophthalmos, AION can occur even in the absence of glaucoma, highlighting the need for increased vigilance regarding the risk of optic nerve ischemia [14]. Additionally, the patient in our case did not undergo surgery; instead, vision improved after conservative treatment. This suggests that for the treatment of nanophthalmos combined with AION, a comprehensive treatment plan that includes improving microcirculation and providing neurotrophic support may be necessary [15].
Nanophthalmos complicated by NA-AION requires meticulous diagnostic differentiation from optic neuritis, as their clinical presentations, including acute vision loss and optic disc edema, show significant overlap. Optic neuritis is a common optic nerve disease, usually associated with infections or autoimmune diseases, and responds well to corticosteroid therapy. Contrast-enhanced MRI can detect optic nerve enhancement. In our case, MRI-T1 with fat suppression showed no enhancement of the optic nerve. FFA revealed typical AION manifestations, such as segmental filling defects of the right optic disc, leading to the diagnosis of AION. In contrast, AION generally has a poor prognosis with limited vision recovery [16]. Therefore, during the diagnostic process, it is essential to comprehensively consider the patient's medical history, clinical manifestations, and imaging results to avoid misdiagnosis [17].
The treatment for our case mainly included corticosteroids, improving microcirculation, and neurotrophic support as conservative therapeutic approaches. However, corticosteroids had limited efficacy in treating AION. The patient's vision further deteriorated after receiving high-dose methylprednisolone sodium succinate. This presentation may represent the natural progression of non-arteritic anterior ischemic optic neuropathy (NA-AION), wherein visual deterioration can occur spontaneously regardless of corticosteroid treatment or standard therapy. Such spontaneous worsening is typically correlated with either the expansion of the initial ischemic insult or secondary axonal damage. Subsequently, the treatment was adjusted to low-dose corticosteroids combined with medications to improve microcirculation and provide neurotrophic support. The patient's vision improved, and the optic disc edema was reduced [18]. The patient is a young adult with no significant comorbidities. Throughout the treatment course, no complications or adverse reactions were observed. This indicates that a comprehensive treatment plan has a positive effect on the patient's condition to some extent. For such rare cases, further clinical observation and research are needed to accumulate more experience and enhance the understanding and management of this condition [19. 20].
Conservative management may be effective in this case; however, long-term continuous monitoring remains crucial. Surgical intervention may be warranted to optimally address anatomically driven AION. This case emphasizes the need for heightened vigilance regarding the possibility of AION when patients with nanophthalmos present with vision loss and optic disc edema to avoid misdiagnosis and mistreatment. Moreover, the uniqueness of this case lies in its rare combination of diseases and partial response to conventional treatment, providing new insights into the risk factors and management strategies for optic nerve disorders in patients with nanophthalmos.
For high-risk populations, particularly those with nanophthalmos, arteritis, hypertension, or diabetes mellitus, comprehensive preventive strategies should be implemented. These include careful management of systemic conditions, such as optimizing blood pressure, controlling glycemic levels, and managing lipids, as well as specific interventions like antithrombotic therapy and immunomodulation when indicated. Regular monitoring and early intervention of these risk factors are crucial for preventing adverse outcomes.
CONCLUSION
Patients with true microphthalmos may face an increased risk of developing AION due to anatomical abnormalities that result in optic nerve hypoperfusion. When these patients with decreased visual acuity and optic disc edema, they should be closely monitored for the potential occurrence of AION to prevent misdiagnosis and mistreatment. Early diagnosis, comprehensive treatment, and long-term follow-up are of utmost importance in improving prognosis. Moreover, given the rarity of such cases, further clinical observation and research are imperative to deepen our understanding and to guide the development of more effective diagnosis and treatment strategies.
Correction notice
None
Acknowledgements
None
Author contributions
(I) Conception and design: Yuying Ji
(II) Administrative support: Jin Ma
(III) Provision of study materials or patients: Yuying Ji
(IV) Collection and assembly of data: Xuelin Chen
(V) Data analysis and interpretation: Yaji Li
(VI) Manuscript writing: All authors
(VII) Final approval of manuscript: All authors
Funding
None
Conflict of interests
None of the authors has any conflicts of interest to disclose.All authors have declared in the completed the ICMJE uniform disclosure form
Patient consent for publication
None
Ethics statement
None
Provenance and peer review
This article was a standard submission to our journal. The article has undergone peer review with our anonymous review system
Data sharing statement
None
Open access statement
This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license).
基金
参考文献
1. Khan AO. Nanophthalmos in children: morphometric and clinical characterization. J AAPOS. 2020, 24(4): 259. DOI: 10.1016/j.jaapos.2020.03.011.
2. Gaier ED, Torun N. The enigma of nonarteritic anterior ischemic optic neuropathy: an update for the comprehensive ophthalmologist. Curr Opin Ophthalmol. 2016, 27(6): 498-504. DOI: 10.1097/ICU.0000000000000318.
3. Vanden Heuvel C, Aldred B, Boulter T, et al. MFRP variant results in nanophthalmos, retinitis pigmentosa, variability in foveal avascular zone. Ophthalmic Genet. 2023, 44(1): 83-88. DOI: 10.1080/13816810.2022.2103835.
4. Tao J, Jin ZB, Shen RJ. MFRP, PRSS56, and MYRF account for 60.5% of a Chinese cohort with nanophthalmos. Clin Exp Ophthalmol. 2025, 53(2): 194-208. DOI: 10.1111/ceo.14465.
5. Garg P, Kumar B, Dubey S. A unique case of bilateral nanophthalmos and pigmentary retinal abnormality with unilateral angle closure glaucoma and optic disc pit. BMC Ophthalmol. 2023, 23(1): 391. DOI: 10.1186/s12886-023-03132-8.
6. Yang N, Zhao LL, Liu J, et al. Nanophthalmos: an update on the biological parameters and fundus abnormalities. J Ophthalmol. 2021, 2021: 8853811. DOI: 10.1155/2021/8853811.
7. Venkatesh R, Mishra P, Nahata H, et al. Retinal Arcades in posterior microphthalmos: biometric correlation. Clin Exp Optom. 2023, 106(6): 619-625. DOI: 10.1080/08164622.2022.2104628.
8. Ahmad M Mansour, Michael W Stewart, Salma W Yassine, Carl-Joe Z Mehanna, Antonio Marcelo B Casella. Unmeasurable small size superficial and deep foveal avascular zone in nanophthalmos: the Collaborative Nanophthalmos OCTA Study. Br J Ophthalmol. 2019, 103(8): 1173-1178. DOI: 10.1136/bjophthalmol-2018-312963.
9. Wen Z, Chen Y, Lin H. OCT angiographic features of choroidal microcirculation in nanophthalmos and its correlation with optic nerve perfusion. Int J Ophthalmol. 2024, 17(5): 821-827. DOI: 10.18240/ijo.2024.05.18.
10. Zhang L, Wang Y, Li J. Relationship between visual function and retinal structure in patients with non-arteritic anterior ischemic optic neuropathy at different disease stages. Chin J Ocul Fundus Dis. 2024, 30(9): 689-694. Document ID: 4718813728.
11. Coleman-Belin J, Harris A, Chen B, et al. Aging effects on optic nerve neurodegeneration. Int J Mol Sci. 2023, 24(3): 2573. DOI: 10.3390/ijms24032573.
12. Mansour AM, Uwaydat SH, Hamam R, et al. Sclerectomy reverses nanophthalmic optic neuropathy. Case Rep Ophthalmol. 2024, 15(1): 284-291. DOI: 10.1159/000537829.
13. Park DH, Park TK, Ohn YH, et al. Linezolid induced retinopathy. Doc Ophthalmol. 2015, 131(3): 237-244. DOI: 10.1007/s10633-015-9518-6.
14. Mollan SP. Semaglutide and nonarteritic anterior ischemic optic neuropathy. JAMA Ophthalmol. 2024, 142(8): 740-741. DOI: 10.1001/jamaophthalmol.2024.2514.
15. Bennett JL, Costello F, Chen JJ, et al. Optic neuritis and autoimmune optic neuropathies: advances in diagnosis and treatment. Lancet Neurol. 2023, 22(1): 89-100. DOI: 10.1016/S1474-4422(22)00187-9.
16. Siriratnam P, Huda S, Butzkueven H, et al. A comprehensive review of the advances in neuromyelitis optica spectrum disorder. Autoimmun Rev. 2023, 22(12): 103465. DOI: 10.1016/j.autrev.2023.103465.
17. Kim NN, Abdel-Mannan O, Davidson J, et al. Leigh syndrome mimicking neuromyelitis optica spectrum disorder (NMOSD). Mult Scler. 2023, 29(7): 889-892. DOI: 10.1177/13524585231172950.
18. Shen ZJ, Shen L, Wang H. Clinical characteristics and surgical treatment of idiopathic uveal effusion syndrome. Int J Ophthalmol. 2022, 15(4): 604-608. DOI: 10.18240/ijo.2022.04.13.
19. Badla O, Badla BA, Almobayed A, et al. Ischemic optic neuropathy: a review of current and potential future pharmacotherapies. Pharmaceuticals (Basel), 2024, 17(10): 1281. DOI: 10.3390/ph17101281.
20. Zhou N, Yang L, Xu X, et al. Uveal effusion syndrome: clinical characteristics, outcome of surgical treatment, and histopathological examination of the sclera. Front Med (Lausanne), 2022, 9: 785444. DOI: 10.3389/fmed.2022.785444.