高效液相色谱法测定曲伏噻吗滴眼液中曲伏前列素有关物质
关键词
摘要
全文
文章亮点
1. 关键发现
· 笔者参考美国药典曲伏前列素质量标准中杂质A检查项色谱条件,优化了色谱柱、进样体积和洗脱程序,并通过分析方法验证,证明该方法可以用于检验曲伏噻吗滴眼液的曲伏前列素有关物质。2. 已知与发现
· 目前文献道的曲伏前列素有关物质检验多采用高效液相色谱法,样品分析长达3小时。笔者参考药典中的方法,建立了一种新的曲伏前列素有关物质检验方法。3. 意义与改变
· 意义:该方法降低了曲伏噻吗滴眼液曲伏前列素有关物质的检验成本和检测时长,提高了检验效率。青光眼主要特征为视神经损害和视野缺损,是致盲率极高的眼病[1-4]。治疗手段主要通过药物控制眼压,预防和延缓病情发展进程[5-6],在临床治疗中,单一药物的治疗常常难以达到理想的效果,因此,青光眼一般采用联合用药[7-9]。
1 材料与方法
1.1 仪器
高效液相色谱仪(Waters Arc,包括四元泵、自动进样器、柱温箱、光电二极管阵列检测器等);BT-125D型电子天平(德国Sartorius公司)。1.2 试剂与样品
曲伏前列素对照品(USP,批号为R078A0,质量浓度为0.507 mg/mL),5,6-反式曲伏前列素对照品(TRC,批号为7-VHP-95-2,质量百分比为96.28%),15-酮曲伏前列素(TRC,批号:3-JPD-149-2,质量百分比为94.91%),曲伏噻吗滴眼液[规格:2.5 mL∶曲伏前列素0.1 mg和马来酸噻吗洛尔12.5 mg(以噻吗洛尔计),批号为22110901、22111101、22111102,中山大学中山眼科中心],曲伏前列素空白辅料滴眼液(批号为220824,中山大学中山眼科中心),乙腈为色谱纯,磷酸和氢氧化钠为分析纯,水为注射用水。1.3 检测方法
采用Agilent SB-C18色谱柱(50 mm×2.1 mm,2.7 μm);以磷酸溶液(取磷酸2.0 mL,加水稀释并定容至1 000 mL,用氢氧化钠溶液调节pH至3.0)为流动相A,乙腈为流动相B,按表1进行梯度洗脱;流速为每分钟3.0 mL;柱温为30 ℃;检测波长为220 nm;进样体积100 μL。
表 1 流动相洗脱梯度
Table 1 Elution gradient of mobile phase
|
时间/min |
流动相A/% |
流动相B/% |
|
0 |
60 |
40 |
|
45 |
60 |
40 |
|
46 |
93 |
7 |
|
55 |
93 |
7 |
|
56 |
60 |
40 |
|
60 |
60 |
40 |
2 结果
2.1系统适用性
图 1 系统适用性色谱图
Figure 1 System suitability chromatograms
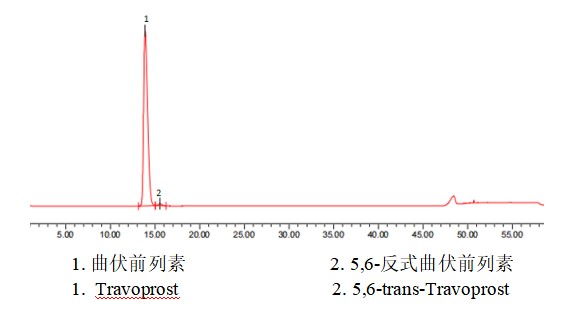
表 2 系统适用性结果
Table 2 Results of system suitability
|
保留时间/min |
相对保留时间 |
分离度 |
理论板数 |
拖尾因子 |
|
|
曲伏前列素 |
13.881 |
1.0 |
/ |
4587 |
1.4 |
|
5,6-反式曲伏前列素 |
15.552 |
1.1 |
2.1 |
6417 |
1.1 |
2.2专属性试验
图 2 专属性色谱图
Figure 2 Specialized chromatogram
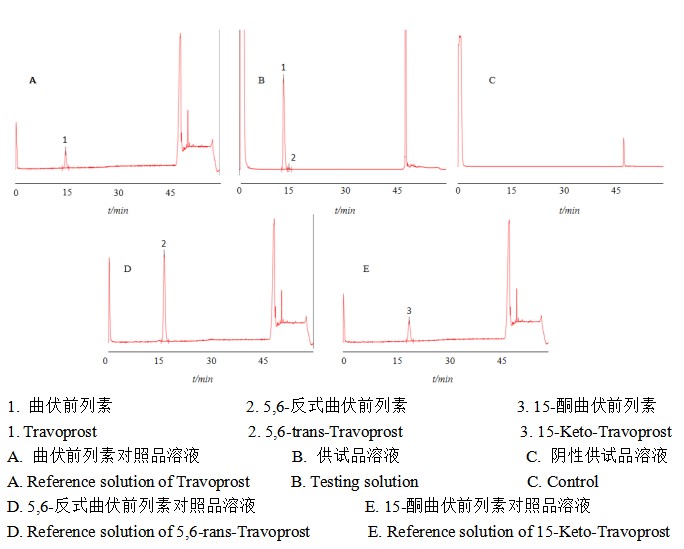
2.3 强制降解试验
表 3 强制降解试验结果
Table 3 Results of forced degradation test
|
未破坏 |
酸破坏 |
碱破坏 |
氧化破坏 |
高温破坏 |
光照破坏 |
|
|
曲伏前列素峰纯度角度 |
0.181 |
0.141 |
0.174 |
0.165 |
0.155 |
0.152 |
|
曲伏前列素峰纯度阈值 |
0.217 |
0.217 |
0.217 |
0.215 |
0.211 |
0.210 |
|
主峰与相邻杂质峰分离度 |
2.1 |
1.9 |
1.8 |
1.7 |
1.5 |
1.6 |
|
5,6-反式曲伏前列素/% |
1.7 |
1.5 |
1.3 |
1.1 |
1.1 |
1.2 |
|
15-酮曲伏前列素/% |
未检出 |
未检出 |
未检出 |
未检出 |
未检出 |
未检出 |
|
未知杂质总和/% |
未检出 |
未检出 |
0.4 |
4.9 |
未检出 |
0.3 |
|
杂质个数 |
1 |
1 |
2 |
7 |
1 |
3 |
|
杂质总量/% |
1.7 |
1.5 |
1.7 |
6.0 |
1.1 |
1.5 |
|
物料平衡/% |
/ |
99.4 |
95.6 |
98.0 |
98.7 |
97.9 |
2.4 检测限与定量限
表 4 检测限和定量限结果
Table 4 Results of etection limit and quantification limit
|
检测限 |
定量限 |
|||
|
信噪比 |
质量浓度/(μg/mL) |
信噪比 |
质量浓度/(μg/mL) |
|
|
曲伏前列素 |
5 |
0.020 |
23 |
0.041 |
|
5,6-反式曲伏前列素 |
5 |
0.020 |
19 |
0.040 |
|
15-酮曲伏前列素 |
5 |
0.020 |
24 |
0.039 |
2.5 线性与范围
表 5 曲伏前列素线性与范围
Table 5 The linear and range of Travoprost
|
溶液号 |
||||||
|
1 |
2 |
3 |
4 |
5 |
6 |
|
|
质量浓度/(μg/mL) |
0.041 |
0.406 |
0.811 |
1.622 |
2.434 |
3.245 |
|
峰面积(A) |
10 907 |
91 310 |
160 343 |
315 385 |
475 727 |
636 094 |
5, 6-反式曲伏前列素线性:以5, 6-反式曲伏前列素(C)为纵坐标,峰面积(A)为横坐标制作标准曲线图,结果如表6所示。
表 6 5, 6- 反式曲伏前列素线性与范围
Table 6 The linear and range of 5, 6-trans-Travoprost
|
1 |
2 |
3 |
4 |
5 |
6 |
|
|
质量浓度/(μg/mL) |
0.040 |
0.404 |
0.807 |
1.614 |
2.422 |
3.229 |
|
峰面积(A) |
7 839 |
78 224 |
150 512 |
301 848 |
449 962 |
602 124 |
15-酮曲伏前列素线性:以15-酮曲伏前列素(C)为纵坐标,峰面积(A)为横坐标制作标准曲线图,结果如表7所示。
表 7 15- 酮曲伏前列素线性与范围
Table 7 The linear and range of 15-Keto-Travoprost
|
1 |
2 |
3 |
4 |
5 |
6 |
|
|
质量浓度/(μg/mL) |
0.039 |
0.386 |
0.772 |
1.544 |
2.316 |
3.088 |
|
峰面积(A) |
9 327 |
104 560 |
211 499 |
421 197 |
630 768 |
850 387 |
因此,5, 6-反式曲伏前列素校正因子为1.05,15-酮曲伏前列素校正因子为0.71。
2.6 重复性
2.7 准确度试验
表 8 5,6- 反式曲伏前列素回收率实验结果
Table 8 Results of adding 5,6-trans-Travoprost recovery
|
测得量/μg |
加入量/μg |
回收率/% |
平均回收率/% |
RSD/% |
|
|
1 |
12.23 |
12.92 |
94.7 |
95.2 |
0.5 |
|
2 |
12.30 |
12.92 |
95.2 |
||
|
3 |
12.26 |
12.92 |
94.9 |
||
|
4 |
15.51 |
16.14 |
96.1 |
||
|
5 |
15.46 |
16.14 |
95.8 |
||
|
6 |
15.41 |
16.14 |
95.5 |
||
|
7 |
18.41 |
19.37 |
95.0 |
||
|
8 |
18.41 |
19.37 |
95.0 |
||
|
9 |
18.39 |
19.37 |
94.9 |
表 9 15- 酮曲伏前列素加样回收率实验结果
Table 9 Results of adding 15-Keto-Travoprost recovery
|
测得量/μg |
加入量/μg |
回收率/% |
平均回收率/% |
RSD/% |
|
|
1 |
2.32 |
2.47 |
93.9 |
92.7 |
1.2 |
|
2 |
2.29 |
2.47 |
92.7 |
||
|
3 |
2.27 |
2.47 |
91.9 |
||
|
4 |
2.85 |
3.09 |
92.2 |
||
|
5 |
2.90 |
3.09 |
93.9 |
||
|
6 |
2.81 |
3.09 |
90.9 |
||
|
7 |
3.47 |
3.71 |
93.5 |
||
|
8 |
3.48 |
3.71 |
93.8 |
||
|
9 |
3.40 |
3.71 |
91.6 |
2.8 溶液稳定性
2.9耐用性试验
表 10 耐用性试验结果
Table 10 Results of durability test
|
变化条件 |
5,6-反式曲伏前列素/% |
15-酮曲伏前列素/% |
其他最大单个杂质/% |
总杂质/% |
|
|
柱温/℃ |
25 |
1.63 |
未检出 |
未检出 |
1.63 |
|
30 |
1.64 |
未检出 |
未检出 |
1.64 |
|
|
35 |
1.62 |
未检出 |
未检出 |
1.62 |
|
|
流速/(mL/min) |
2.5 |
1.66 |
未检出 |
未检出 |
1.66 |
|
3.0 |
1.64 |
未检出 |
未检出 |
1.64 |
|
|
3.5 |
1.62 |
未检出 |
未检出 |
1.62 |
|
|
流动相pH |
2.7 |
1.67 |
未检出 |
未检出 |
1.67 |
|
3.0 |
1.64 |
未检出 |
未检出 |
1.64 |
|
|
3.3 |
1.67 |
未检出 |
未检出 |
1.67 |
|
|
色谱柱批号 |
B23131 |
1.64 |
未检出 |
未检出 |
1.64 |
|
B22782 |
1.63 |
未检出 |
未检出 |
1.63 |
2.10 样品含量测定
表 11 三批曲伏噻吗滴眼液的曲伏前列素有关物质结果
Table 11 Results of related substances in three batches of Trivoxetine eye drops
|
5,6-反式曲伏前列素/% |
15-酮曲伏前列素/% |
其他最大单个杂质/% |
曲伏前列素总杂质/% |
|
|
22110901 |
1.74 |
未检出 |
未检出 |
1.74 |
|
22111101 |
1.69 |
未检出 |
未检出 |
1.69 |
|
22111102 |
1.72 |
未检出 |
未检出 |
1.72 |
3 讨论
表 12 分析方法对比
Table 12 Comparison of analysis methods
|
新建标准 |
USP43标准 |
《高效液相色谱法测定曲伏噻吗滴眼液中曲伏前列素有关物质含量》中方法 |
||||||||||||||||||||||||||||||||||||||||||||||||||
|
分析方法 |
高效液相色谱法 |
高效液相色谱法 |
高效液相色谱法 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
色谱柱 |
Agilent SB-C18色谱柱(50 mm×2.1 mm,2.7 mm) |
150 mm×4.6 mm,5 mm,packing L1 |
Phenomenex LUNA 苯基己基色谱柱(3.0 mm×150 mm, 3 μm) |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
流动相 |
以磷酸溶液(取磷酸2.0 mL,加水稀释并定容至1 000 mL,用氢氧化钠试液调节pH至3.0)为流动相A,乙腈为流动相B |
缓冲盐溶液(2.18 mg/mL-辛烷磺酸钠水溶液,用磷酸调节pH值至3.5)-乙腈(33:17) |
流动相A为20 mmol/L庚烷磺酸钠溶液(用磷酸调pH值至2.5),流动相B为甲醇,流动相C为乙腈 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
洗脱程序 |
|
等度洗脱 |
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
流速/(mL/min) |
3.0 |
2.0 |
0.5 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
柱温/℃ |
30 |
/ |
40 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
检测波长/nm |
220 |
220 |
220 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
进样体积/μL |
100 |
100 |
100 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
系统适用性要求 |
理论板数按曲伏前列素峰计算不低于1 500,拖尾因子不大于2.0,曲伏前列素峰与5,6-反式曲伏前列素峰的相对保留时间分别为1.0和1.1,其分离度应大于1.5 |
曲伏前列素峰峰面积RSD不大于2.0%,曲伏前列素峰与5,6-反式曲伏前列素峰的相对保留时间分别为1.0和1.1,其分离度应大于1.5 |
曲伏前列素及各杂质均能有效分析,分离度均大于1.5 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
对照品溶液/(μg/mL) |
0.4 |
40 |
0.4 |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
供试品溶液/(μg/mL) |
40 |
40 |
40 |
声明
在论文撰写中无使用生成式人工智能。论文撰写中的所有内容均由作者独立完成,并对出版物的真实性和准确性承担全部责任。利益冲突
所有作者均声明不存在利益冲突。开放获取声明
基金
参考文献
Chen SR, Lai MY, Lin PJ. Pathogenesis and treatment of neovascular glaucoma[J]. Int Eye Sci, 2024, 24(10): 1605-1609.
Wei SJ, Yang JH. Research progress of glaucoma diagnosis in myopic patients[J]. Anhui Med Pharm J, 2024, 28(10): 1912-1917. DOI: 10.3969/j.issn.1009-6469.2024.10.002.
Liu QQ, Zhao SY, Wang P, et al. Relationship between self-perceived burden and mental resilience in patients with glaucoma[J]. Psychologies Magazine, 2024, 19(16): 22-24+28. DOI: 10.19738/j.cnki.psy.2024.16.006.
Zhuo YH, Wu J. Application potential and direction of artificial intelligence in glaucoma prevention and treatment[J]. Chin J Ophthalmol, 2023, 59(09): 691-695. DOI: 10.3760/cma.j.cn112142-20230519-00204.
He M, Tang L. Application progress of telemedicine in the management of glaucoma diagnosis and treatment[J]. J Otolaryngol Ophthalmol Shandong Univ, 2023, 37(4): 187-195. DOI: 10.6040/j.issn.1673-3770.0.2022.135.
Mu W, Ding YZ, Xi YF, et al. Progress in research and application of local anti-glaucoma drugs[J]. Chin J New Drugs Clin Remedies, 2024, 43(8): 574-581. DOI: 10.14109/j.cnki.xyylc.2024.08.03.
Mao HY, Xiong K, Tang LW, et al. Selection of initial treatment methods for primary angle closure glaucoma[J]. Recent Adv Ophthalmol, 2024, 44(5): 405-409. DOI: 10.13389/j.cnki.rao.2024.0079.
Gan P, Liu XK, Li X. Research progress on novel intraocular pressure lowering drugs using trabecular meshwork pathway[J]. World Clin Drug, 2024, 45(1): 77-83. DOI: 10.13683/j.wph.2024.01.013.
Lin QX, Zhang L, Li YH. Comparison of curative effects of travoprost eye drops and timolol maleate eye drops in the treatment of primary open angle glaucoma[J]. Chin J Clin Ration Drug Use, 2023, 16(30): 149-152. DOI: 10.15887/j.cnki.13-1389/r.2023.30.042.
Xu N, Ni SH, Zhao J, et al. Meta-analysis of efficacy between Travoprost and Timolol in treating primary open angle glaucoma[J]. Int Eye Sci, 2022, 22(4): 616-622.
Li SA, Peng L, Sun Y. Effect observation of travoprost on patients with primary open-angle glaucoma[J]. Qingdao Med J, 2022, 54(1): 35-37.
Zhang L. Effects of travoprost eye drops on hemodynamics and visual function in patients with primary open-angle glaucoma[J]. J N Pharm, 2022, 19(2): 190-193. DOI: 10.3969/j.issn.1672-8351.2022.02.062.
Zhou J. Analysis of the clinical efficacy of travoprost eye drops in the treatment of patients with primary open angle glaucoma[J]. Contemp Med, 2021, 27(24): 81-83.
Kang K, Guo M, Zou JY, et al. Application of travor prostaglandin eye drops in primary open angle glaucoma[J]. Heilongjiang Med Pharm, 2021, 44(3): 92-93. DOI: 10.3969/j.issn.1008-0104.2021.03.042.
Han BY, Mi Q, Jia GM, et al. Comparison of the therapeutic effects of latanoprost, timolol and travoprost in treatment of primary open angle glaucoma[J]. Hebei Med J, 2020, 42(9): 1324-1327. DOI: 10.3969/j.issn.1002-7386.2020.09.009.
Tang JP. Clinical efficacy of travoprost eye drops in the treatment of primary open angle glaucoma[J]. Chin J Clin Ration Drug Use, 2020, 13(22): 74-75. DOI: 10.15887/j.cnki.13-1389/r.2020.22.032.
Fei LH, Fei HY, Wu J. Determination of the related substances of travoprost in travoprost and timolol maleate eye drops by HPLC[J]. Her Med, 2023, 42(3): 411-415. DOI: 10.3870/j.issn.1004-0781.2023.03.022.
Guo CC, Li DP, Li T, et al. Trevoloproic acidImpurity content determination of travoprost eye dropsby HPLC method[J]. Chem Eng, 2019, 33(10): 32-34. DOI: 10.16247/j.cnki.23-1171/tq.20191032.
Chen JT, Li YJ, Weng SM. Determination of travoprost content in travoprost eye drops by high performance liquid chromatography[J]. Yan Ke Xue Bao, 2024, 39 (12): 600-607. DOI:10.12419/24093001.
Song W. Interpretation of the guiding principles of analytical method validation based on the Chinese pharmacopoeia 2020 version[J]. Shandong Chem Ind, 2021, 50(13): 95-96. DOI: 10.19319/j.cnki.issn.1008-021x.2021.13.040.
Xia ZH, Liu WZ, Han XR, et al. Re-elaboration on the questions of HPLC analytical method validation[J]. Pharm Clin Res, 2020, 28(2): 120-124. DOI: 10.13664/j.cnki.pcr.2020.02.011.
Liu Y, Yue JF. Drug inspection technology[M]. 2nd ed. Beijing: Chemical Industry Press, 2024.
Zhu RD, Geng Y, Tan DJ, et al. Statistical evaluation of method validation for quantitative analysis of impurites[J]. Chin J Pharm Anal, 2019, 39(2): 215-222. DOI: 10.16155/j.0254-1793.2019.02.05.
Tan DJ, Zhu RD, Geng Y, et al. Analysis of the current situation of domestic and foreign guidelines for method validation[J]. Chin J Pharm Anal, 2019, 39(2): 191-195. DOI: 10.16155/j.0254-1793.2019.02.01.
Wang SH, Wu Y, Wang Y, et al. Discussion on the main points of drug analytical method validation[J]. Chin J Pharm Anal, 2018, 38(9): 1646-1651. DOI: 10.16155/j.0254-1793.2018.09.25.